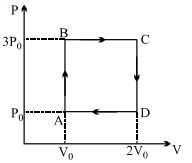
A ideal monoatomic gas is carried around the cycle $ABCDA$ as shown in the fig. The efficiency of the gas cycle is

Advanced
Heat is absorbed only during processes $AB$ and $BC$
Gas is monoatomic.Hence $C_{v}=\frac{3}{2} R, C_{p}=\frac{5}{2} R$
Heat absorbed during $\mathrm{AB}, \Delta Q_{A B}=n C_{v} \Delta T=\frac{3}{2} n R \Delta T=\frac{3}{2} V \Delta P=3 P_{o} V_{o}$
Heat absorbed during $\mathrm{BC}, \Delta Q_{B C}=n C_{p} \Delta T=\frac{5}{2} n R \Delta T=\frac{5}{2} P \Delta V=\frac{15}{2} P_{o} V_{o}$
Hence, net heat absorbed $=\Delta Q=\Delta Q_{A B}+\Delta Q_{B C}=\frac{21}{2} P_{o} V_{o}$
Also, net work done $=\Delta W$ =area under $P-V$ graph $=2 P_{o} V_{o}$
Hence efficiency $=\frac{\Delta W}{\Delta Q}=\frac{4}{21}$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1In a thermodynamics process, pressure of a fixed mass of a gas is changed in such a manner that the gas releases $20 J$ of heat and $8J$ of work is done on the gas. If the initial internal energy of the gas was $30J.$ The final internal energy will be ...... $J$View Solution
- 2One mole of an ideal gas undergoes a cyclic process, consisting of two isochores and two isobars. Temperature at $1$ and $3$ equal to $T_1$ and $T_3$ respectively. The work done by the gas over the cycle, if the point $2$ and $4$ lie on the same isothermView Solution
- 3In thermodynamic process, $200$ Joules of heat is given to a gas and $100$ Joules of work is also done on it. The change in internal energy of the gas is ........ $J$View Solution
- 4A sample of an ideal gas undergoes an isothermal expansion. If $d Q, d U$ and $d W$ represent the amount of heat supplied, the change in internal energy and the work done respectively, thenView Solution
- 5Let $\eta_{1}$ is the efficiency of an engine at $T _{1}=447^{\circ}\,C$ and $T _{2}=147^{\circ}\,C$ while $\eta_{2}$ is the efficiency at $T _{1}=947^{\circ}\,C$ and $T _{2}=47^{\circ}\,C$. The ratio $\frac{\eta_{1}}{\eta_{2}}$ will be.View Solution
- 6In a thermodynamic process two moles of a monatomic ideal gas obeys $P \propto V^{-2}$. If temperature of the gas increases from $300 \,K$ to $400 \,K$, then find work done by the gas .........$R$ (where $R=$ universal gas constant).View Solution
- 7View SolutionWhen heat in given to a gas in an isobaric process, then
- 8$Assertion :$ In isothermal process whole of the heat supplied to the body is converted into internal energy.View Solution
$Reason :$ According to the first law of thermodynamics : $\Delta Q = \Delta U + p\Delta V$ - 9View SolutionIn isothermal expansion, the pressure is determined by
- 10During an experiment, an ideal gas is found to obey a condition $\frac{{{P^2}}}{\rho }$ = constant [$\rho =$ density of the gas]. The gas is initially at temperature $T,$ pressure $P$ and density $\rho$ . The gas expands such that density changes to $\rho/2$View Solution
