A measure of the degree of disorder of a system is known as
Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1In a mechanical refrigerator, the low temperature coils are at a temperature of $-23°C$ and the compressed gas in the condenser has a temperature of $27°C.$ The theoretical coefficient of performance isView Solution
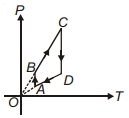
- 2A $P-T$ graph is shown for a cyclic process. Select correct statement regarding thisView Solution
- 3During which of the following thermodynamic process represented by $P V$ diagram the heat energy absorbed by system may be equal to area under $P V$ graph?View Solution
- 4View SolutionThe internal energy of the gas increases In
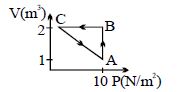
- 5An ideal gas is taken through the cycle $A \to B \to C \to A$ , as shown in the figure. If the net heat supplied to the gas in the cycle is $5\ J$, the work done by the gas in the process $C \to A$ is .... $J$View Solution
- 6One mole of an ideal monoatomic gas is heated at a constant pressure of one atmosphere from ${0^o}C$ to ${100^o}C$. Then the change in the internal energy isView Solution
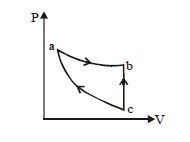
- 7An ideal gas expands isothermally along $ab$ and does $600\,J$ of work. During the processView Solution
- 8In a carnot engine, the temperature of reservoir is $527^{\circ} C$ and that of $\operatorname{sink}$ is $200 \; K$. If the workdone by the engine when it transfers heat from reservoir to sink is $12000 \; kJ$, the quantity of heat absorbed by the engine from reservoir is $\times 10^{6} \; J$View Solution
- 9For an adiabatic expansion of an ideal gas, the fractional change in its pressure is equal to (where $\gamma$ is the ratio of specific heats):View Solution
- 10The pressure and volume of an ideal gas are related as $\mathrm{PV}^{3 / 2}=\mathrm{K}$ (Constant). The work done when the gas is taken from state $A\left(P_1, V_1, T_1\right)$ to state $\mathrm{B}\left(\mathrm{P}_2, \mathrm{~V}_2, \mathrm{~T}_2\right)$ is :View Solution
