In a carnot engine, the temperature of reservoir is $527^{\circ} C$ and that of $\operatorname{sink}$ is $200 \; K$. If the workdone by the engine when it transfers heat from reservoir to sink is $12000 \; kJ$, the quantity of heat absorbed by the engine from reservoir is $\times 10^{6} \; J$
JEE MAIN 2022, Medium
$\left(T_{2}\right) T_{\sin k}=200 \; K$
$\left(T_{1}\right) T_{\text {Reservoir }}=527+273=800 \; K$
$W=12000 K J=12 \times 10^{6} \; J$
$Q_{1}=$ ?
$\eta=1-\frac{T_{2}}{T_{1}}=\frac{W}{Q_{1}}=1-\frac{200}{800}=\frac{12 \times 10^{6}}{Q_{1}}$
$\frac{3}{4}=\frac{12 \times 10^{6}}{Q_{1}}=Q_{1}=16 \times 10^{6} J$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionWhich relation is correct for isometric process
- 2View SolutionThe efficiency of a Carnot engine depends upon
- 3A gas may expand either adiabatically or isothermally. A number of $P-V$ curves are drawn for the two processes over different range of pressure and volume. It will be found thatView Solution
- 4View SolutionThe adiabatic Bulk modulus of a diatomic gas at atmospheric pressure is
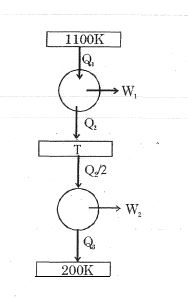
- 5Consider the given series combination of carnot cycles. If $W_1 = W_2$ then the value of $T$ is ...... $K$ (all temperatures are maintained at their respective values)View Solution
- 6View SolutionIn the following figures heat is absorbed by the gas
- 7Avessel with open mouth contains air at $60^oC$. When the vessel is heated upto temperature $T$, one fourth of the air goes out. The value of $T$ is ..... $^oC$View Solution
- 8A sample of gas at temperature $\mathrm{T}$ is adiabatically expanded to double its volume. Adiabatic constant for the gas is $\gamma=3 / 2$. The work done by the gas in the process is : $(\mu=1 \mathrm{~mole})$View Solution
- 9Work done by $0.1$ mole of a gas at ${27^o}C$ to double its volume at constant pressure is ....... $cal$ ($R = 2 \,cal\, mol^{-1}$ $^oC^{-1}$)View Solution
- 10The amount of work done in an adiabatic expansion from temperature $T$ to ${T_1}$ isView Solution
