An ideal gas is taken through the cycle $A \to B \to C \to A$ , as shown in the figure. If the net heat supplied to the gas in the cycle is $5\ J$, the work done by the gas in the process $C \to A$ is .... $J$
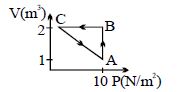

Medium
$\mathrm{dR}=\mathrm{dU}+\mathrm{d} \mathrm{W}$
$5=0+10(2-1)+0+\mathrm{W}_{\mathrm{CA}}$
$\mathrm{W}_{\mathrm{CA}}=5-10$
$=-5$ Joule
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A sample of ideal monoatomic gas is taken round the cycle $ABCA$ as shown in the figure. The work done during the cycle isView Solution
- 2View SolutionThe slopes of isothermal and adiabatic curves are related as
- 3An ideal gas expands in such a way that $PV^2 =$ constant throughout the process.View Solution
- 4View SolutionTwo gases are said to be in thermal equilibrium when they have same
- 5Following figure shows two processes $A$ and $B$ for a gas. If $\Delta Q_A$ and $\Delta Q_B$ are the amount of heat absorbed by the system in two cases, and $\Delta U_A$ and $\Delta U_B$ are changes in internal energies, respectively, thenView Solution
- 6The figure shows $P-V$ diagram of a thermodynamic cycle. Which corresponding curve is correct?View Solution
- 7If ${C_V} = 4.96cal/mole\, K$, then increase in internalenergy when temperature of $2$ moles of this gas is increased from $340 K$ to $342 K$ ....... $cal$View Solution
- 8Which of the accompanying $PV$, diagrams best represents an isothermal processView Solution
- 9View SolutionThe temperature-entropy diagram of a reversible engine cycle is given in the figure. Its efficiency is
- 10If $R =$ universal gas constant, the amount of heat needed to raise the temperature of $2$ mole of an ideal monoatomic gas from $273K$ to $373K$ when no work is done ...... $R$View Solution
