Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A sample of ideal gas $(\gamma = 1.4)$ is heated at constant pressure. If an $100\, J$ is supplied to gas, the work done by the gas is ...... $J$View Solution
- 2View SolutionThe door of a working refrigerator is left open in a well insulated room. The temperature of air in the room will
- 3A gas is suddenly compressed to one fourth of its original volume. What will be its final pressure, if its initial pressure is $P$View Solution
- 4$100\ g$ of water is heated from $30^o\ C$ to $50^o\ C$ Ignoring the slight expansion of the water, the change in its internal energy is ...... $kJ$ (specific heat of water is $4184\ J/kg/K$)View Solution
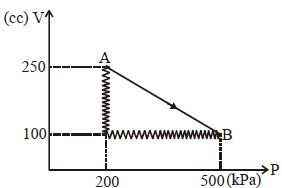
- 5One mole of an ideal gas at $27^{\circ} {C}$ is taken from ${A}$ to ${B}$ as shown in the given ${PV}$ indicator diagram. The work done by the system will be $......\times 10^{-1} \,{J}$View Solution
[Given : $R=8.3\, {J} /\,mole\,{K}, \ln 2=0.6931$ ] (Round off to the nearest integer)
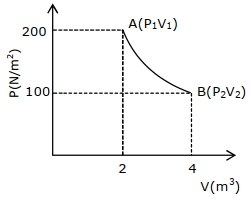
- 6One mole of a perfect gas in a cylinder fitted with a piston has a pressure $P,$ volume $V$ and temperature $T.$ If the temperature is increased by $1 \,K$ keeping pressure constant, the increase in volume isView Solution
- 7The heat energy required to raise the temperature of $5\,moles$ of an ideal gas to $5\,K$ at constant pressure is $600\,J$ . How much heat (in $J$ ) is required to raise the same mass of the same gas to $5\,K$ at constant volume ? (Take $R = 8.3\,J/mole-^oK$ )View Solution
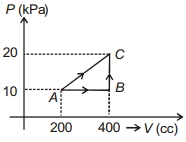
- 8If a gas is taken from $A$ to $C$ through $B$ then heat absorbed by the gas is $8 \,J$. Heat absorbed by the gas in taking it from $A$ to $C$ directly is ............. $J$View Solution
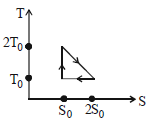
- 9View SolutionThe temperature-entropy diagram of a reversible engine cycle is given in the figure. Its efficiency is
- 10View SolutionWhich of the following parameters does not characterize the thermodynamic state of matter