A monoatomic gas is taken through a process $TP^{-1/3} =$ constant. If heat is given to the gas
Diffcult
$\mathrm{TP}^{-1 / 3}=$ constant
$\mathrm{PVP}^{-1 / 3}=$ constant
$\mathrm{PV}^{3 / 2}=$ constant
$\Rightarrow x=\frac{3}{2}, \gamma=\frac{5}{3}$
$\mathrm{C}=\frac{\mathrm{R}}{\gamma-1}+\frac{\mathrm{R}}{1-\mathrm{x}}=\frac{3 \mathrm{R}}{2}+\frac{\mathrm{R}}{1-\frac{3}{2}}=-\frac{\mathrm{R}}{2}$
$\mathrm{C}$ is negative $\Delta Q=n C \Delta T$
$\Rightarrow$ if $\Delta Q$ is $+v e, \Delta T$ is$ -$ve
$\Rightarrow$ temperature will decrease
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A Carnot engine absorbs an amount $Q$ of heat from a reservoir at an absolute temperature $T$ and rejects heat to a sink at a temperature of $T/3$ . The amount of heat rejected isView Solution
- 2A Carnot's engine working between $400\, K$ and $800\, K$ has a work output of $1200\, J$ per cycle. The amount of heat energy supplied to the engine from the source in each cycle is ........... $J$View Solution
- 3A Carnot engine operating between two reservoirs has efficiency $\frac{1}{3}$. When the temperature of cold reservoir raised by $x$, its efficiency decreases to $\frac{1}{6}$. The value of $x$, if the temperature of hot reservoir is $99^{\circ}\,C$, will be $........\,K$View Solution
- 4View SolutionWhen a gas expands adiabatically
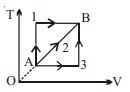
- 5A given mass of a gas expands from a state $A$ to the state $B$ by three paths $1, 2$ and $3$ as shown in $T-V$ indicator diagram. If $W_1, W_2$ and $W_3$ respectively be the work done by the gas along the three paths, thenView Solution
- 6View SolutionFor free expansion of the gas which of the following is true
- 7A mixture of gases at $STP$ for which $\gamma=1.5$ is suddenly compressed to $\frac{1}{9}$ th of its original volume. The final temperature of mixture is .......... $^{\circ} C$View Solution
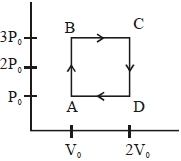
- 8An engine operates by taking a monatomic ideal gas through the cycle shown in the figure. The percentage efficiency of the engine is close to $.......\%$View Solution
- 9During the adiabatic expansion of $2 \,moles$ of a gas, the internal energy was found to have decreased by $100 J$. The work done by the gas in this process is ..... $J$View Solution
- 10Initial pressure and volume of a gas are $P$ and $V$ respectively. First it is expanded isothermally to volume $4V$ and then compressed adiabatically to volume $V$ . The final pressure of gas will be (given $\gamma = 3/2$ )View Solution
