Initial pressure and volume of a gas are $P$ and $V$ respectively. First it is expanded isothermally to volume $4V$ and then compressed adiabatically to volume $V$ . The final pressure of gas will be (given $\gamma = 3/2$ )
Medium
In isothermal process $\mathrm{P}_{1} \mathrm{V}_{1}=\mathrm{P}_{2} \mathrm{V}_{2}$
$\Rightarrow \mathrm{PV}=\mathrm{P}_{2} \times 4 \mathrm{V} \quad \because \mathrm{P}_{2}=\mathrm{P} / 4$
In adiabatic process
$\mathrm{P}_{2} \mathrm{V}_{2}^{\gamma}=\mathrm{P}_{3} \mathrm{V}_{3}^{\gamma} \Rightarrow \frac{\mathrm{P}}{4} \times(4 \mathrm{V})^{15}=\mathrm{P}_{3} \mathrm{V}^{15} \Rightarrow \mathrm{P}_{3}=2 \mathrm{P}$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
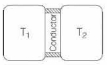
- 1A thin piece of thermal conductor of constant thermal conductivity insulated on the lateral sides connects two reservoirs which are maintained at temperatures $T_{1}$ and $T_{2}$ as shown in the figure alongside. Assuming that the system is in steady state, which of the following plots best represents the dependence of the rate of change of entropy on the ratio of $T_{1} / T_{2}$ ?View Solution
- 2The amount of work done in an adiabatic expansion from temperature $T$ to ${T_1}$ isView Solution
- 3The temperature of sink of Carnot engine is ${27^o}C$. Efficiency of engine is $25\%.$ Then temperature of source is ...... $^oC$View Solution
- 4A reversible engine has an efficiency of $\frac{1}{4}$. If the temperature of the sink is reduced by $58^{\circ} {C}$, its efficiency becomes double. Calculate the temperature of the sink. (In $^{\circ} {C}$)View Solution
- 5A gas is suddenly compressed to one fourth of its original volume. What will be its final pressure, if its initial pressure is $P$View Solution
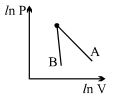
- 6The figure, shows the graph of logarithmic reading of pressure and volume for two ideal gases $A$ and $B$ undergoing adiabatic process. From figure it can be concluded thatView Solution
- 7The volume $V$ of a given mass of monoatomic gas changes with temperature $T$ according to the relation $V = KT ^{2 / 3}$. The workdone when temperature changes by $90\, K$ will be $x\,R$. The value of $x$ is $[ R =$ universal gas constant $]$View Solution
- 8The specific heat at constant pressure of a real gas obeying $\mathrm{PV}^2=\mathrm{RT}$ equation is :View Solution
- 9View SolutionWhich of the following statements is correct for any thermodynamic system
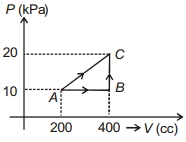
- 10If a gas is taken from $A$ to $C$ through $B$ then heat absorbed by the gas is $8 \,J$. Heat absorbed by the gas in taking it from $A$ to $C$ directly is ............. $J$View Solution