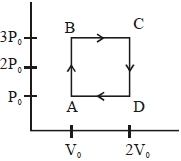
An engine operates by taking a monatomic ideal gas through the cycle shown in the figure. The percentage efficiency of the engine is close to $.......\%$

JEE MAIN 2020, Diffcult
$W _{ ABCDA }=2 P _{0} V _{0}$
$Q_{\text {in }}=Q_{A B}+Q_{B C}$
$Q _{ AB }= nC \left( T _{ B }- T _{ A }\right)$
$=\frac{ n 3 R }{2}\left( T _{ B }- T _{ A }\right)$
$=\frac{3}{2}\left(P_{B} V_{B}-P_{A} V_{A}\right)$
$=\frac{3}{2}\left(3 P_{B} V_{0}=P_{0} V_{0}\right)=3 P_{0} V_{0}$
$Q _{ BC }= nC _{ P }\left( T _{ C }- T _{ B }\right)$
$=\frac{ n 5 R }{2}\left( T _{ C }- T _{ B }\right)$
$=\frac{5}{2}\left(P_{C} V_{C}-P_{B} V_{B}\right)$
$=\frac{5}{2}\left(6 P _{0} V _{0}-3 P _{0} V _{0}\right)=\frac{15}{2} P _{0} V _{0}$
$\eta=\frac{ W }{ Q _{ in }} \times 100=\frac{2 P _{0} V _{0}}{3 P _{0} V _{0}+\frac{15}{2} P _{0} V _{0}} \times 100$
$\eta=\frac{400}{21}=19.04 \approx 19$
$\eta=19$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A diatomic ideal gas undergoes a thermodynamic change according to the $P-V$ diagram shown in the figure. The heat given the gas in $AB$View Solution
- 2The $P-V$ graph of an ideal gas cycle is shown here as below. The adiabatic process is described byView Solution
- 3An ideal gas is made to undergo a cycle depicted by the $p-V$ diagram given below. The curved line from $A$ to $B$ is an adiabat.Then,View Solution
- 4Density $vs$ volume graph is shown in the figure. Find corresponding pressure $vs$ temperature graphView Solution
- 5During an adiabatic process, the pressure of a gas is found to be proportional to the cube of its temperature. The ratio of $\frac{{{C_P}}}{{{C_V}}}$ for the gas isView Solution
- 6A thermodynamic system is taken from state $A$ to $B$ along $ACB$ and is brought back to $A$ along $BDA$ as shown in the $PV$ diagram. The net work done during the complete cycle is given by the areaView Solution
- 7The temperature of a hypothetical gas increases to $\sqrt 2 $ times when compressed adiabatically to half the volume. Its equation can be written asView Solution
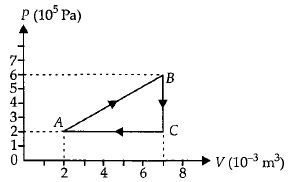
- 8A gas is taken through the cycle $A\to B\to C\to A$ as shown. What is the net work done by the gas ...... $J$ $?$View Solution
- 9View SolutionA cycle tyre bursts suddenly. This represents an
- 10View SolutionIn the following figures heat is absorbed by the gas
