A narrow glass tube, $80 \,cm$ long and opens at both ends, is half immersed in mercury, now the top of the tube is closed and is taken out of mercury. A column of mercury $20 \,cm$ long remains in the tube. Find atmospheric pressure
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A fix amount of nitrogen gas ($1$ mole) is taken and is subjected to pressure and temperature variation. The experiment is performed at high pressure as well as high temperatures. The results obtained are shown in the figures. The correct variation of $PV/RT$ with $P$ will be exhibited byView Solution
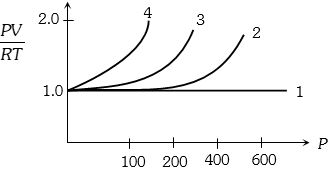
- 2By what factor the $r.m.s.$ velocity will change, if the temperature is raised from $27^\circ C$ to $327^\circ C$View Solution
- 3View SolutionA piston is slowly pushed into a metal cylinder containing an ideal gas. Which of the following statements is/are incorrect?
- 4This question has Statement $- 1$ and Statement $-2$. Of the four choices given after the Statements, choose the one that best describes the two Statements.View Solution
Statement $1$ : The internal energy of a perfect gas is entirely kinetic and depends only on absolute temperature of the gas and not on its pressure or volume.
Statement $2$ : A perfect gas is heated keeping pressure constant and later at constant volume. For the same amount of heat the temperature of the gas at constant pressure is lower than that at constant volume. - 5If $7\, gm N _{2}$ is mixed with $20\, gm$ $Ar$, there $C _{ p } / C _{ v }$ of mixture will beView Solution
- 6View SolutionAn ideal gas is filled in a closed container and container is moving with uniform acceleration in horizontal direction. Neglect gravity. Pressure inside the container is ...............
- 7A thermally insulated vessel contains an ideal gas of molecular mass $M$ and ratio of specific heats $1.4$. Vessel is moving with speed $v$ and is suddenly brought to rest. Assuming no heat is lost to the surrounding and vessel temperature of the gas increases by ... ( $R =$ universal gas constant )View Solution
- 8A resistance coil connected to an external battery is placed inside an adiabatic cylinder fitted with a frictionless pistn and containing an ideal gas. A current $i$ flows through the coil which has a resistance $R$. At what speed must the piston move upward in order that the temperature of the gas remains uchanged? Neglect atmospheric pressure.View Solution
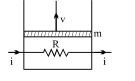
- 9View SolutionIndicator diagram is a ...........
- 10A closed cylindrical vessel contains $N$ moles of an ideal diatomic gas at a temperature $T$. On supplying heat, temperature remains same, but $n$ moles get dissociated into atoms. The heat supplied is .........View Solution
