A fix amount of nitrogen gas ($1$ mole) is taken and is subjected to pressure and temperature variation. The experiment is performed at high pressure as well as high temperatures. The results obtained are shown in the figures. The correct variation of $PV/RT$ with $P$ will be exhibited by
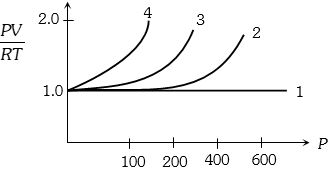

Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionTwo vessels having equal volume contains molecular hydrogen at one atmosphere and helium at two atmospheres respectively. If both samples are at the same temperature, the mean velocity of hydrogen molecules is
- 2Let $\gamma_1$ be the ratio of molar specific heat at constant pressure and molar specific heat at constant volume of a monoatomic gas and $\gamma_2$ be the similar ratio of diatomic gas. Considering the diatomic gas molecule as a rigid rotator, the ratio, $\frac{\gamma_1}{\gamma_2}$ isView Solution
- 3An ideal gas initially at pressure $1$ bar is being compressed from $30 \,m ^{3}$ to $10\, m ^{3}$ volume and its temperature decreases from $320\, K$ to $280\, K$. then find final pressure of gas (in bar)View Solution
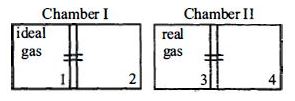
- 4There are two identical chambers, completely thermally insulated from surroundings. Both chambers have a partition wall dividing the chambers in two compartments. Compartment $1$ is filled with an ideal gas and Compartment $3$ is filled with a real gas. Compartments $2$ and $4$ are vacuum . A small hole (orifice) is made in the partition walls and the gases are allowed to expand in vacuumView Solution
Statement $-1$ : No change in the temperature of the gas takes place when ideal gas expands in vacuum. However, the temperature of real gas goes down (cooling) when it expands in vacuum
Statement $-2$ : The internal energy of an ideal gas is only kinetic. The internal energy of a real gas is kinetic as well as potential
- 5View SolutionThe temperature below which a gas should be cooled, before it can be liquified by pressure only is termed as
- 6View SolutionThe relation between two specific heats of a gas is
- 7Two moles of ideal helium gas are in a rubber balloon at $30^o C$. The balloon is fully expandable and can be assumed to required no energy in its expansion. The temperature of the gas in the balloon is slowly changed to $35^o C$. The amount of heat required in raising the temperature is nearly...... $J$View Solution
(take $R$ $=$ $8.31$ $J/mol.K$) - 8On $0^\circ C$ pressure measured by barometer is $760\, mm.$ What will be pressure on $100^\circ C$View Solution
- 9Two thermally insulated vessels $1$ and $2$ are filled with air at temperatures $({T_1},\,\,{T_2}),$ volume $({V_1},\,\,{V_2})$ and pressure $({P_1},\,\,{P_2})$ respectively. If the valve joining the two vessels is opened, the temperature inside the vessel at equilibrium will beView Solution
- 10$Assertion :$ Air pressure in a car tyre increases during driving.View Solution
$Reason :$ Absolute zero temperature is not zero energy temperature.
