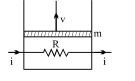
A resistance coil connected to an external battery is placed inside an adiabatic cylinder fitted with a frictionless pistn and containing an ideal gas. A current $i$ flows through the coil which has a resistance $R$. At what speed must the piston move upward in order that the temperature of the gas remains uchanged? Neglect atmospheric pressure.

Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A diatomic gas of molecular mass $40 \,g / mol$ is filled in a rigid container at temperature $30^{\circ} C$. It is moving with velocity $200 \,m / s$. If it is suddenly stopped, the rise in the temperature of the gas is .........View Solution
- 2Five moles of helium are mixed with two moles of hydrogen to form a mixture. Take molar mass of helium $M_1=4\ g$ and that of hydrogen $M_2=2\ g$ If the internal energy of He sample of $100\,\,J$ and that of the hydrogen sample is $200\,\,J$, then the internal energy of the mixture is ..... $J$View Solution
- 3View SolutionSelect the incorrect statement about ideal gas.
- 4If pressure of $C{O_2}$ (real gas) in a container is given by $P = \frac{{RT}}{{2V - b}} - \frac{a}{{4{b^2}}}$ then mass of the gas in container is ...... $gm$View Solution
- 5The molar specific heats of an ideal gas at constant pressure and volume are denoted by $C _{ P }$ and $C _{ V }$ respectively. If $\gamma=\frac{C_{p}}{C_{V}}$ and $R$ is the universal gas constant, then $C _{ V }$ is equal toView Solution
- 6One litre of oxygen at a pressure of $1\, atm$ and two litres of nitrogen at a pressure of $0.5\, atm$ are introduced into a vessel of volume $1L$. If there is no change in temperature, the final pressure of the mixture of gas (in $atm$) isView Solution
- 7A gas obeying the equation of state $p V=R T$ undergoes a hypothetical reversible process described by the equation, $p V^{5 / 3} \exp \left(-\frac{p V}{E_{0}}\right)=C_{1}$, where $C_{1}$ and $E_{0}$ are dimensioned constants. Then, for this process, the thermal compressibility at high temperatureView Solution
- 8$70$ calories of heat are required to raise the temperature of $2$ moles of an ideal gas at constant pressure from $30°C$ to $35°C.$ The amount of heat required to raise the temperature of same gas through the same range $(30°C$ to $35°C)$ at constant volume ..... $cal$ $(R = 2 \,cal/mol/K)$View Solution
- 9A pressure cooker contains air at $1$ atm and $30^o C$. If the safety value of the cooler blows when the inside pressure $ \ge 3$ atm, then the maximum temperature of the air, inside the cooker can be .... $^oC$View Solution
- 10At constant temperature on increasing the pressure of a gas by $5\%$ will decrease its volume by ..... $\%$View Solution
