One mole of an ideal gas with $\gamma = 1.4$, is adiabatically compressed so that its temperature rises from $27°C$ to $35°C$. The change in the internal energy of the gas is ....... $J$ $(R = 8.3\,J/mol.K)$
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1The gas law $\frac{{PV}}{T} = $ constant is true forView Solution
- 2View SolutionWhen heat is given to a gas in an isothermal change, the result will be
- 3A cylinder with a movable piston contains $3\,moles$ of hydrogen at standard temperature and pressure. The walls of the cylinder are made of a heat insulator, and the piston is insulated by having a pile of sand on it. By what factor does the pressure of the gas increases if the gas is compressed to half its original volume?View Solution
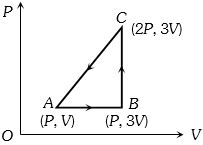
- 4A sample of an ideal gas is taken through a cycle a shown in figure. It absorbs $50J$ of energy during the process $AB$, no heat during $BC$, rejects $70J$ during $CA.$ $40J$ of work is done on the gas during $BC$. Internal energy of gas at $A$ is $1500J$, the internal energy at $C$ would be ........ $J$View Solution
- 5$110\; J$ of heat is added to a gaseous system, whose internal energy change is $40\; J$, then the amount of external work done is ........ $J$View Solution
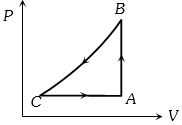
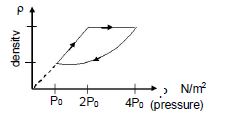
- 6An ideal gas undergoes cyclic process as shown in density pressure graph. During the process $AB$ the work done $|W_{AB}| = 70\,J$ . During the process $BC$, the gas absorbs $150\,J$ of heat. During the process $CA$ , gas undergoes expansion and does $210\,J$ of workView Solution
- 7A gas is compressed at a constant pressure of $50N/{m^2}$ from a volume of $10{m^3}$ to a volume of $4{m^3}$. Energy of $100 J$ then added to the gas by heating. Its internal energy isView Solution
- 8View SolutionTemperature is a measurement of coldness or hotness of an object. This definition is based on
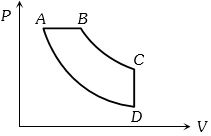
- 9View SolutionIn pressure-volume diagram given below, the isochoric, isothermal, and isobaric parts respectively, are
- 10An ideal gas is taken around $ABCA$ as shown in the above $P-V$ diagram. The work done during a cycle isView Solution