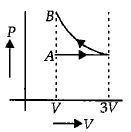
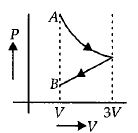
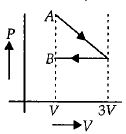
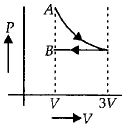
One mole of an ideal gas goes from an initial state $A$ to final state $B$ via two processes : It first undergoes isothermal expansion from volume $V$ to $3\, V$ and then its volume is reduced from $3\, V$ to $V$ at constant pressure. The correct $P-V$ diagram representing the two processes is
AIPMT 2012, Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1Match List$-I$ with List$-II$View Solution
List$-I$ List$-II$ $(a)$ Isothermal $(i)$ Pressure constant $(b)$ Isochoric $(ii)$ Temperature constant $(c)$ Adiabatic $(iii)$ Volume constant $(d)$ Isobaric $(iv)$ Heat content is constant Choose the correct answer from the options given below
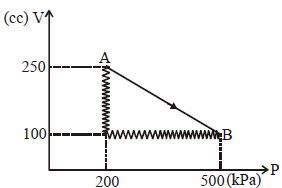
- 2A monoatomic gas is taken along path $AB$ as shown. Calculate change in internal energy of systemView Solution
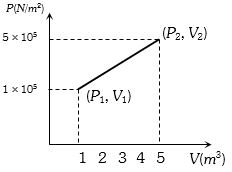
- 3A system changes from the state $({P_1},\;{V_1})$ to $({P_2},{V_2})$ as shown in the figure. What is the work done by the systemView Solution
- 4The temperature inside and outside a refrigerator are $273 \,K$ and $300 \,K$ respectively. Assuming that the refrigerator cycle is reversible, for every joule of work done, the heat delivered to the surrounding will be nearly ........ $J$View Solution
- 5A Carnot engine works first between $200^{\circ} C$ and $0^{\circ} C$ and then between $0^{\circ} C$ and $-200^{\circ} C$. The ratio of its efficiency in the two cases isView Solution
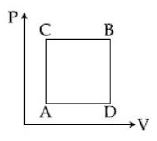
- 6A gas can be taken from $A$ to $B$ via two different processes $ACB$ and $ADB$. When path $ACB$ is used $60\, J$ of heat flows into the system and $30\, J$ of work is done by the system. If path $ADB$ is used work down by the system is $10\, J$. the heat flow into the system in path $ADB$ is ..... $J$View Solution
- 7A gas is compressed from a volume of $2\,m^3$ to a volume of $1\, m^3$ at a constant pressure of $100\, N/m^2$. Then it is heated at constant volume by supplying $150\, J$ of energy. As a result, the internal energy of the gasView Solution
- 8View SolutionWhich of the following processes is reversible
- 9A Carnot engine absorbs an amount $Q$ of heat from a reservoir at an abosolute temperature $T$ and rejects heat to a sink at a temperature of $T/3.$ The amount of heat rejected isView Solution
- 10An ideal gas is expanded adiabatically at an initial temperature of $300 K$ so that its volume is doubled. The final temperature of the hydrogen gas is $(\gamma = 1.40)$View Solution