A reversible heat engine converts one-fourth of the heat input into work. When the temperature of the sink is reduced by $52\, K$, its efficiency is doubled. The temperature in Kelvin of the source will be ...... .
JEE MAIN 2021, Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1$5.6$ $liter$ of helium gas at $STP$ is adiabatically compressed to $0.7$ $liter$. Taking the initial temperature to be $T_1$, the work done in the process isView Solution
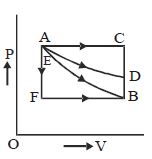
- 2An ideal system can be brought from state $A$ to $B$ through four paths as shown in the figure. The energy given to the system is minimum inView Solution
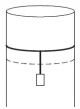
- 3A long cylindrical pipe of radius $20 \,cm$ is closed at its upper end and has an airtight piston of negligible mass as shown. When a $50 \,kg$ mass is attached to the other end of the piston, it moves down. If the air in the enclosure is cooled from temperature $T$ to $T-\Delta T$, the piston moves back to its original position. Then $\Delta T / T$ is close to (Assuming air to be an ideal gas, $g=10 \,m / s ^2$, atmospheric pressure is $10^5 \,Pa$ )View Solution
- 4A Carnot engine operates between ${227^o}C$ and ${27^o}C.$ Efficiency of the engine will beView Solution
- 5Given below are two statements. One is labelled as Assertion A and the other is labelled as Reason $R$.View Solution
Assertion $A$ : If $dQ$ and $dW$ represent the heat supplied to the system and the work done on the system respectively. Then according to the first law of thermodynamics $d Q=d U-d W$.
Reason $R :$ First law of thermodynamics is based on law of conservation of energy.
In the light of the above statements, choose the correct answer from the option given below :
- 6In an adiabatic process $90J$ of work is done on the gas. The change in internal energy of the gas is ....... $J$View Solution
- 7Work done by air when it expands from $50\, litres$ to $150\, litres$ at a constant pressure of $2$ atmosphere isView Solution
- 8If one mole of an ideal gas goes through the process $A \rightarrow B$ and $B \rightarrow C .$ Given that $T _{ A }=400\, K ,$ and $T _{ C }=400 \,K .$ If $\frac{ P _{ B }}{ P _{ A }}=\frac{1}{5},$ then find the heat supplied to the gas (in $J$)View Solution
- 9Which of the following graphs correctly represents the variation of $\beta = - (dV/dP)/V$ with $P$ for an ideal gas at constant temperatureView Solution
- 10The first law of thermodynamics can be written as $ \Delta U = \Delta Q + \Delta W$ for an ideal gas. Which of the following statements is correct?View Solution
