Which of the following graphs correctly represents the variation of $\beta = - (dV/dP)/V$ with $P$ for an ideal gas at constant temperature
Medium
(a) For an isothermal process $PV =$ constant
==> $PdV + VdP = 0$ ==> $ - \frac{1}{V}\left( {\frac{{dV}}{{dP}}} \right) = \frac{1}{P}$
So, $\beta = \frac{1}{P}$ graph will be rectangular hyperbola.
==> $PdV + VdP = 0$ ==> $ - \frac{1}{V}\left( {\frac{{dV}}{{dP}}} \right) = \frac{1}{P}$
So, $\beta = \frac{1}{P}$ graph will be rectangular hyperbola.
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1An ideal heat engine working between temperature $T_1$ and $T_2 $ has an efficiency $\eta$, the new efficiency if both the source and sink temperature are doubled, will beView Solution
- 2In an adiabatic process where in pressure is increased by $\frac{2}{3}\% $ if $\frac{{{C_p}}}{{{C_v}}} = \frac{3}{2},$ then the volume decreases by aboutView Solution
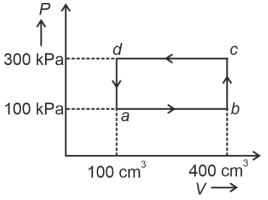
- 3A cyclic process of a thermodynamic system is taken through $a$ $b$ $c$ $d$ $a$. The work done by the gas along the path $b$ $c$ isView Solution
- 4$300 \,cal$. of heat is given to a heat engine and it rejects $225 \,cal$. of heat. If source temperature is $227^{\circ} C$, then the temperature of sink will be____${ }^{\circ} C$.View Solution
- 5Areversible adiabatic path on a $P-V$ diagram for an ideal gas passes through stateAwhere $P=0$.$7\times 10^5 \,\,N/ m^{-2}$ and $v = 0.0049 \,\,m^3$. The ratio of specific heat of the gas is $1.4$. The slope of path at $A$ is :View Solution
- 6View SolutionThe door of a working refrigerator is left open in a well insulated room. The temperature of air in the room will
- 7$V\, = \,K\,{\left( {\frac{P}{T}} \right)^{0.33}}$ where $k$ is constant. It is an,View Solution
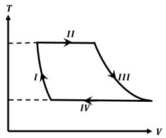
- 8One mole of a monatomic ideal gas undergoes a cyclic process as shown in the figure (where $V$ is the volume and $T$ is the temperature). Which of the statements below is (are) true?View Solution
(image)
$(A)$ Process $I$ is an isochoric process $(B)$ In process $II$, gas absorbs heat
$(C)$ In process $IV$, gas releases heat $(D)$ Processes $I$ and $III$ are $not$ isobaric
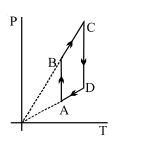
- 9View SolutionPressure versus temperature graph of an ideal gas is shown in figure
- 10A heat engine operates with the cold reservoir at temperature $324 K$. The minimum temperature of the hot reservoir, if the heat engine takes $300 \; J$ heat from the hot reservoir and delivers $180 \; J$ heat to the cold reservoir per cycle, is $\dots \; K .$View Solution
