Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1In an $H_2$ gas process, $PV^2 =$ constant. The ratio of work done by gas to change in its internal energy isView Solution
- 2A diatomic ideal gas is compressed adiabatically to $\frac{1}{32}$ of its initial volume. If the initial temperature of the gas is $T_1$ (in Kelvin) and the final temperature is $a T_1$, the value of $a$ isView Solution
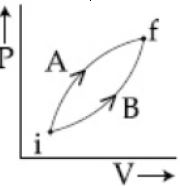
- 3Following figure shows two processes $A$ and $B$ for a gas. If $\Delta Q_A$ and $\Delta Q_B$ are the amount of heat absorbed by the system in two cases, and $\Delta U_A$ and $\Delta U_B$ are changes in internal energies, respectively, thenView Solution
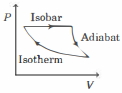
- 4The efficiency of the cycle shown below in the figure (consisting of one isobar, one adiabat and one isotherm) is $50 \%$. The ratio $x$, between the highest and lowest temperatures attained in this cycle obeys (the working substance is an ideal gas)View Solution
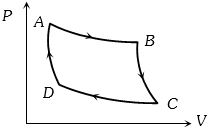
- 5The $P-V$ graph of an ideal gas cycle is shown here as below. The adiabatic process is described byView Solution
- 6A gas is suddenly compressed to $1/4$ th of its original volume at normal temperature. The increase in its temperature is ....... $K$ $(\gamma = 1.5)$View Solution
- 7Two identical samples of a gas are allowed to expand $(i)$ isothermally $(ii)$ adiabatically. Work done isView Solution
- 8An enclosed one mole of a monoatomic gas is taken through a process $A$ to $B$ as shown in figure. The molar heat capacity of the gas for this process isView Solution
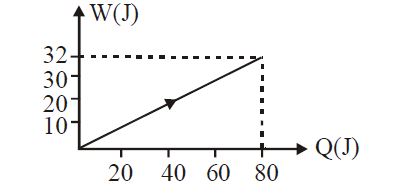
- 9View SolutionIn the figure shown, amount of heat supplied to one mole of an ideal gas is plotted on the horizontal axis and amount of work done by gas is drawn on vertical axis. Assuming process be isobaric i.e. gas can be
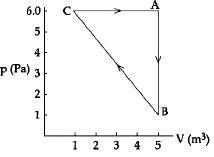
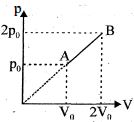
- 10For the given cyclic process $CAB$ as shown for a gas, the work done is ..... $J$View Solution