A sample of gas expands from volume ${V_1}$ to ${V_2}$. The amount of work done by the gas is greatest when the expansion is
AIIMS 1998,AIPMT 1997, Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1The work of $146\,kJ$ is performed in order to compress one kilo mole of a gas adiabatically and in this process the temperature of the gas increases by $7\,^oC$ . The gas is $(R = 8.3\, J\, mol^{-1}\, K^{-1})$View Solution
- 2Two cylinders $A$ and $B$ of equal capacity are connected to each other via a stop cock. A contains an Ideal gas at standard temperature and pressure. $B$ is completely evacuated. The entire system is thermally insulated. The stop cock is suddenly opened. The process is :View Solution
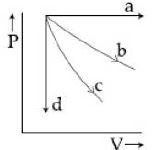
- 3View SolutionThe given diagram shows four processes i.e., isochoric, isobaric, isothermal and adiabatic. The correct assignment of the processes, in the same order is given by
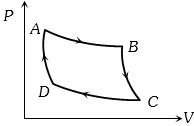
- 4Carnot cycle (reversible) of a gas represented by a Pressure-Volume curve is shown in the diagramConsider the following statementsView Solution
$I.$ Area $ABCD =$ Work done on the gas
$II.$ Area $ABCD =$ Net heat absorbed
$III.$ Change in the internal energy in cycle $= 0$
Which of these are correct
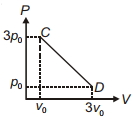
- 5The process $C D$ is shown in the diagram. As system is taken from $C$ to $D$, what happens to the temperature of the system?View Solution
- 6$5.6$ liter of helium gas at $STP$ is adiabatically compressed to $0.7$ liter. Taking the initial temperature to be $T _1$, the work done in the process isView Solution
- 7View SolutionWhich of the following process will give maximum amount of heat to surrounding when volume becomes half of initial
- 8A sample of $1$ mole gas at temperature $\mathrm{T}$ is adiabatically expanded to double its volume. If adiabatic constant for the gas is $\gamma=\frac{3}{2}$, then the work done by the gas in the process is:View Solution
- 9An electric heater supplies heat to a system at a rate of $100\;W.$ If system performs work at a rate of $75$ Joules per second. At what rate (in $J/s$) is the internal energy increasing?View Solution
- 10Initial pressure and volume of a gas are $P$ and $V$ respectively. First it is expanded isothermally to volume $4V$ and then compressed adiabatically to volume $V$ . The final pressure of gas will be (given $\gamma = 3/2$ )View Solution
