Which of the following process will give maximum amount of heat to surrounding when volume becomes half of initial
Medium
Here isochoric process is not possible and for
adiabatic process, $\mathrm{Q}=0$ For isothermal process
${Q_{rejected}}\, = - W = nR{T_0}\ell n\left( {{V_0}/\frac{{{V_0}}}{2}} \right) = nR{T_0}\ell n\,2$
$=0.693 \mathrm{nRT}_{0}$
For isobaric process
$Q_{\text {rejected }}=-n C_{p} \Delta T=-n\left(\frac{f}{2}+1\right) n R\left(-\frac{T_{0}}{2}\right)$
$=\left(0.5+\frac{f}{4}\right) n R T_{0}>0.693 n R T_{0}$
It is clear that more heat is rejected in isobaric process.
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A refrigerator works between $4^o C$ and $30^o C.$ It is required to remove $600$ calories of heat every second in order to keep the temperature of the refrigerated space constant. The power required is ....... $W$ (Take $1\, cal \,=\, 4.2\, Joules\,)$View Solution
- 2$A$ reversible engine converts one-sixth of the heat input into work. When the temperature of the sink is reduced by $ 62^oC$, the efficiency of the engine is doubled. The temperatures of the source and sink areView Solution
- 3An ideal Carnot engine, whose efficiency is $40 \%$ receives heat at $500\; K$. If its efficiency is $50 \%$ then the intake temperature for the same exhaust temperature is ......... $K$View Solution
- 4One mole of an ideal gas is taken through an adiabatic process where the temperature rises from $27^{\circ} {C}$ to $37^{\circ} {C}$. If the ideal gas is composed of polyatomic molecule that has $4$ vibrational modes which of the following is true?View Solution
- 5View SolutionWhich of the following laws of thermodynamics defines the term internal energy?
- 6A diatomic ideal gas is used in a carnot engine as the working substance. If during the adiabatic expansion part of the cycle the volume of the gas increases from $V$ to $32\,V$ , the efficiency of the engine isView Solution
- 7The process $AB$ is shown in the diagram. As the gas is taken from $A$ to $B$, its temperatureView Solution
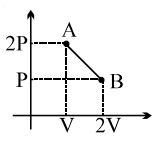
- 8View SolutionThe thermodynamic process, in which internal energy of the system remains constant is ...........
- 9One mole of ideal gas undergoes a linear process as shown in the figure below. Its temperature expressed as a function of volume $V$ isView Solution
- 10A thermodynamic process is the pressure and volumes corresponding to some points in the figure are, $P_A = 3 \times 10^4 Pa$, $V_A = 2 \times 10^{-3}\, m^3$, $P_B = 8 \times 10^4 Pa$, $V_D = 5 \times 10^{-3}\,m^3$. In process $AB, 600\, J$ of heat and in process $BC, 200\, J$ of heat is added to the system. The change in the internal energy in process $AC$ would be .... $J$View Solution