$\Rightarrow \mathrm{T}(\mathrm{V})^{\frac{3}{2}-1}=\mathrm{T}_{\mathrm{f}}(2 \mathrm{~V})^{\frac{3}{2}-1}$
$\Rightarrow \mathrm{TV}^{\frac{1}{2}}=\mathrm{T}_{\mathrm{f}}(2)^{\frac{1}{2}}(\mathrm{~V})^{\frac{1}{2}}$
$\Rightarrow \mathrm{T}_{\mathrm{f}}=\left(\frac{\mathrm{T}}{\sqrt{2}}\right)$
$\text { Now, W.D. }=\frac{\mathrm{nR} \Delta \mathrm{T}}{1-\gamma}=\frac{1 \cdot \mathrm{R}\left[\frac{\mathrm{T}}{\sqrt{2}}-\mathrm{T}\right]}{1-\frac{3}{2}}$
$\Rightarrow \text { W.D. }=2 \mathrm{RT}\left[1-\frac{1}{\sqrt{2}}\right]$
$\Rightarrow \text { W.D. }=\mathrm{RT}[2-\sqrt{2}]$
Download our appand get started for free
Similar Questions
- 1An ideal gas expands isothermally from a volume ${V_1}$ to ${V_2}$ and then compressed to original volume ${V_1}$adiabatically. Initial pressure is ${P_1}$ and final pressure is ${P_3}$. The total work done is $W$. ThenView Solution
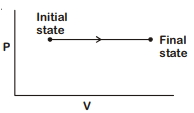
- 2The $P-V$ diagram for an ideal gas in a piston cylinder assembly undergoing a thermodynamic process is shown in the figure. The process isView Solution
- 3Consider $1 \,kg$ of liquid water undergoing change in phase to water vapour at $100^{\circ} C$. At $100^{\circ} C$, the vapour pressure is $1.01 \times 10^5 \,N - m ^2$ and the latent heat of vaporization is $22.6 \times 10^5 \,Jkg ^{-1}$. The density of liquid water is $10^3 \,kg m ^{-3}$ and that of vapour is $\frac{1}{1.8} \,kg m ^{-3}$. The change in internal energy in this phase change is nearly ............ $\,J kg ^{-1}$View Solution
- 4In an isochoric process if ${T_1} = {27^o}C$ and ${T_2} = {127^o}C,$ then ${P_1}/{P_2}$ will be equal toView Solution
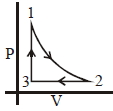
- 5Which of the following is an equivalent cyclic process corresponding to the thermodynamic cyclic given in the figure? where, $1 \rightarrow 2$ is adiabatic.View Solution
(Graphs are schematic and are not to scale)
- 6A mixture of ideal gas containing $5$ moles of monatomic gas and $1$ mole of rigid diatomic gas is initially at pressure $P _0$, volume $V _0$ and temperature $T _0$. If the gas mixture is adiabatically compressed to a volume $V _0 / 4$, then the correct statement(s) is/are,View Solution
(Give $2^{1.2}=2.3 ; 2^{3.2}=9.2 ; R$ is gas constant)
$(1)$ The final pressure of the gas mixture after compression is in between $9 P _0$ and $10 P _0$
$(2)$ The average kinetic energy of the gas mixture after compression is in between $18 RT _0$ and $19 RT _0$
$(3)$ The work $| W |$ done during the process is $13 RT _0$
$(4)$ Adiabatic constant of the gas mixture is $1.6$
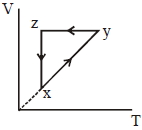
- 7A thermodynamic cycle $xyzx$ is shown on a $V-T$ diagram.View Solution
The $P-V$ diagram that best describes this cycle is
(Diagrams are schematic and not to scale)
- 8In changing the state of thermodynamics from $A$ to $B$ state, the heat required is $Q$ and the work done by the system is $W.$ The change in its internal energy isView Solution
- 9A balloon filled with helium $\left(32^{\circ} C \right.$ and $1.7\; atm$.) bursts. Immediately afterwards the expansion of helium can be considered asView Solution
- 10$Assertion :$ Air quickly leaking out of a balloon becomes cooler.View Solution
$Reason :$ The leaking air undergoes adiabatic expansion.
