The work of $146\,kJ$ is performed in order to compress one kilo mole of a gas adiabatically and in this process the temperature of the gas increases by $7\,^oC$ . The gas is $(R = 8.3\, J\, mol^{-1}\, K^{-1})$
Medium
$w=\frac{n R \Delta T}{\gamma-1}$
$146 \times 10^{3}=\frac{10^{3} \times 8.3 \times 7}{\gamma-1}$
$\gamma-1=\frac{8.3 \times 7}{146}=\frac{58.1}{146}$
$\gamma=\frac{58}{146}+1$
$\gamma=\frac{58+146}{146}$
$=\frac{204}{146}$
$=\frac{7}{5}$ diatomic nature.
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionA reversible engine and an irreversible engine are working between the same temperatures. The efficiency of the ...........
- 2View SolutionDuring the thermodynamic process shown in figure for an ideal gas
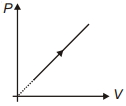
- 3$V\, = \,K\,{\left( {\frac{P}{T}} \right)^{0.33}}$ where $k$ is constant. It is an,View Solution
- 4View SolutionThe efficiency of a Carnot engine depends upon
- 5A mixture of gases at $STP$ for which $\gamma=1.5$ is suddenly compressed to $\frac{1}{9}$ th of its original volume. The final temperature of mixture is .......... $^{\circ} C$View Solution
- 6View SolutionWork done in the cyclic process shown in figure is ...........
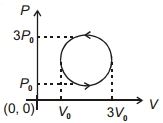
- 7When an ideal gas in a cylinder was compressed isothermally by a piston, the work done on the gas was found to be $1.5 \times {10^4}\;joules$. During this process aboutView Solution
- 8Two different adiabatic paths for the same gas intersect two isothermal curves as shown in$P-V$ diagram. The relation between the ratio $\frac{V_a}{V_d}$ and the ratio $\frac{V_b}{V_c}$ is:View Solution
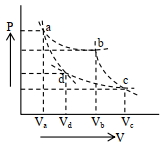
- 9A gas taken through cyclic process $ABCA$ is shown in figure. If $2.4\,cal.$ of heat is given in the process, what is value of $J$ ...... $J/cal$ (mechanical equivalent of heat)?View Solution
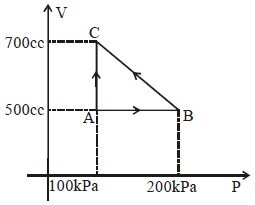
- 10A mixture of gases at $STP$ for which $\gamma=1.5$ is suddenly compressed to $\frac{1}{9}$ th of its original volume. The final temperature of mixture is .......... $^{\circ} C$View Solution
