Heat is given to an ideal gas in an isothermal process.
$A.$ Internal energy of the gas will decrease.
$B.$ Internal energy of the gas will increase.
$C.$ Internal energy of the gas will not change.
$D.$ The gas will do positive work.
$E.$ The gas will do negative work.
Choose the correct answer from the options given below :
JEE MAIN 2023, Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1Water falls from a height of $60\,m$ at the rate of $15\,kg/s$ to operate a turbine. The losses due to frictional forces are $10\%$ of energy. How much power is generated by the turbine ....... $kW$ $(g = 10\,m/s^2)$View Solution
- 2View SolutionIn practice, all heat engines have efficiency less than that of a Carnot engine because
- 3View SolutionWhich is incorrect
- 4A thermodynamic system goes from states $(i)\,\,{P_1}, V$ to $2{P_1}, V\, (ii)\, P, V$ to $P, 2V.$ Then work done in the two cases isView Solution
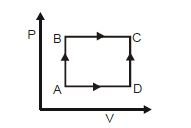
- 5A shown in the figure, the amount of heat absorbed along the path $ABC$ is $90\,J$ and the amount of work done by the system is $30\,J$ . If the amount of work done along the path $ADC$ is $20\,J$ , the amount of heat absorbed will be .... $J$View Solution
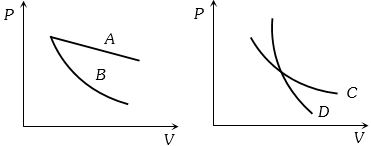
- 6In the following figure, four curves $A, B, C$ and $D$ are shown. The curves areView Solution
- 7A given ideal gas with $\gamma = \frac{{{C_p}}}{{{C_v}}} = 1.5$ at a temperature $T$. If the gas is compressed adiabatically to one-fourth of its initial volume, the final temperature will be ..... $T$View Solution
- 8The amount of heat needed to raise the temperature of $4\, moles$ of a rigid diatomic gas from $0^{\circ} {C}$ to $50^{\circ} {C}$ when no work is done is ......${R}$ ($R$ is the universal gas constant)View Solution
- 9One mole of ${O_2}$ gas having a volume equal to $22.4$ litres at ${0^o}C$ and $1$ atmospheric pressure in compressed isothermally so that its volume reduces to $11.2$ litres. The work done in this process is ...... $J$View Solution
- 10View SolutionIf a cylinder containing a gas at high pressure explodes, the gas undergoes
