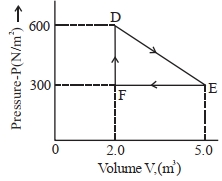
A thermodynamic system is taken from an original state $D$ to an intermediate state $E$ by the linear process shown in the figure. Its volume is then reduced to the original volume from $E$ to $F$ by an isobaric process. The total work done by the gas from $D$ to $E$ to $F$ will be $......J$

JEE MAIN 2022, Medium
$W _{ DE }=\frac{1}{2}(600+300) 3\,J$
$=1350\,J$
$W _{ EF }=-300 \times 3=-900\,J$
$W _{ DEF }=450\,J$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1One gm mol of a diatomic gas $(\gamma = 1.4)$ is compressed adiabatically so that its temperature rises from ${27^o}C$ to ${127^o}C$. The work done will beView Solution
- 2View SolutionCompressed air in the tube of a wheel of a cycle at normal temperature suddenly starts coming out from a puncture. The air inside
- 3$Assertion :$ When a glass of hot milk is placed in a room and allowed to cool, its entropy decreases.View Solution
$Reason :$ Allowing hot object to cool does not violate the second law of thermodynamics. - 4In a thermodynamics process, pressure of a fixed mass of a gas is changed in such a manner that the gas releases $20\,J$ of heat and $8\,J$ of work is done on the gas. If the initial internal energy of the gas was $30\,J$. The final internal energy will be...... $J$View Solution
- 5If one mole of an ideal gas goes through the process $A \rightarrow B$ and $B \rightarrow C .$ Given that $T _{ A }=400\, K ,$ and $T _{ C }=400 \,K .$ If $\frac{ P _{ B }}{ P _{ A }}=\frac{1}{5},$ then find the heat supplied to the gas (in $J$)View Solution
- 6Determine coefficient of performance of given temperature limit.View Solution
$T_{1}=27^{\circ} C$ [outside fridge]
$T_{2}=-23^{\circ} C$ [inside fridge]
- 7An ideal gas at atmospheric pressure is adiabatically compressed so that its density becomes $32$ times of its initial value. If the final pressure of gas is $128$ atmosphers, the value of $\gamma$ the gas isView Solution
- 8An insulator container contains $4\, moles$ of an ideal diatomic gas at temperature $T.$ Heat $Q$ is supplied to this gas, due to which $2 \,moles$ of the gas are dissociated into atoms but temperature of the gas remains constant. ThenView Solution
- 9A thermodynamic process in which temperature $T$ of the system remains constant though other variable $P$ and $V$ may change, is calledView Solution
- 10Consider a spherical shell of radius $R$ at temperature $T$. The black body radiation inside it can be considered as an ideal gas of photons with internal energy per unit volume$E=$ $\frac{U}{V} \propto {T^4}$ and pressure $P = \frac{1}{3}\left( {\frac{U}{V}} \right)$ If the shell now undergoes an adiabatic expansion the relation between $T$ and $R$ isView Solution
