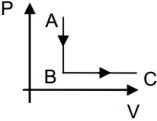
If one mole of an ideal gas goes through the process $A \rightarrow B$ and $B \rightarrow C .$ Given that $T _{ A }=400\, K ,$ and $T _{ C }=400 \,K .$ If $\frac{ P _{ B }}{ P _{ A }}=\frac{1}{5},$ then find the heat supplied to the gas (in $J$)

AIIMS 2019, Medium
The expression to calculate the heat supply to the gas is give as,
$\Delta Q = n \Delta T \left( C _{ p }- C _{ v }\right)$
Substitute the values.
$\Delta Q = n \Delta T \left( C _{ p }- C _{ v }\right)$
$=1 \times 8.31 \times\left(400-\frac{400}{5}\right)$
$=2659.2 J$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A monatomic gas at pressure $P_1$ and volume $V_1$ is compressed adiabatically to ${\frac{1}{8}}^{th}$ of its original volume. What is the final pressure of the gas is ........ $P_1$?View Solution
- 2View SolutionThe temperature-entropy diagram of a reversible engine cycle is given in the figure. Its efficiency is
- 3A diatomic ideal gas undergoes a thermodynamic change according to the $P-V$ diagram shown in the figure. The heat given the gas in $AB$View Solution
- 4$Assertion :$ The isothermal curves intersect each other at a certain point.View Solution
$Reason :$ The isothermal change takes place slowly, so, the isothermal curves have very little slope. - 5For one complete cycle of a thermodynamic process on a gas as shown in the $P-V$ diagram, Which of following is correctView Solution
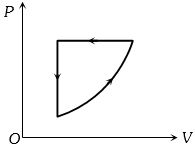
- 6View SolutionIn which thermodynamic process, volume remains same
- 7An ideal gas is subjected to a thermodynamic process $PV^{2.5} = 0.40$ where $P$ is in $Pa$ and $V$ is in $m^3$.What is the slope of the $P-V$ curve with volume plotted against $x-$ axis at $V=1\, m^3$ ?View Solution
- 8In the diagrams $(i)$ to $(iv)$ of variation of volume with changing pressure is shown. A gas is taken along the path $ABCD. $ The change in internal energy of the gas will beView Solution
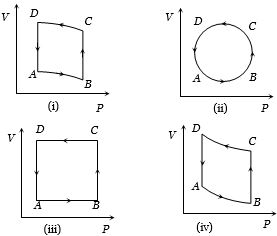
- 9View SolutionWhen an ideal diatomic gas is heated at constant pressure, the fraction of the heat energy supplied which increases the internal energy of the gas, is
- 10An ideal gas undergoes a four step cycle as shown in the $P-V$ diagram below. During this cycle, in which step heat is absorbed by the gasView Solution