A thermodynamic process in which temperature $T$ of the system remains constant though other variable $P$ and $V$ may change, is called
Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionWhich relation is correct for isometric process
- 2An ideal gas, initially in state $\left( P _{12}, V _1, T _1\right)$ is expanded isobarically to $\left( P _{12}, V _2, T _2\right)$, then adiabatically $\left( P _{34}, V _3, T _3\right)$. It is then contracted isobarically to $\left( P _{34}, V _4, T _4\right)$ and finally adiabatically back to the initial state. The efficiency of this cycle isView Solution
- 3How much work to be done in decreasing the volume of and ideal gas by an amount of $2.4 \times {10^{ - 4}}{m^3}$ at normal temperature and constant normal pressure of .......$joule$ $1 \times {10^5}N/{m^2}$View Solution
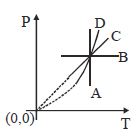
- 4Following figure shows $P-T$ graph for four processes $A, B, C$ and $D$. Select the correct alternativeView Solution
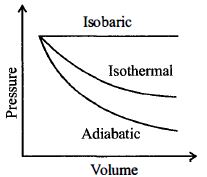
- 5A sample of gas expands from $V_1$ to $V _2$. In which of the following, the work done will be greatest ?View Solution
- 6A Carnot engine works between $27^{\circ} C$ and $127^{\circ} C$. Heat supplied by the source is $500\, J$. then heat ejected to the sink is (in $J$)View Solution
- 7A polyatomic gas $\left( {\gamma = \frac{4}{3}} \right)$ is compressed to $\frac{1}{8}$ of its volume adiabatically. If its initial pressure is ${P_o}$, its new pressure will beView Solution
- 8An ideal gas is subjected to an isothermal expansion such that its volume changes from $V_i$ to $V_f$ and pressure from $P_i$ to $P_f$. The work done on the gas is :View Solution
- 9A sample of gas expands from volume ${V_1}$ to ${V_2}$. The amount of work done by the gas is greatest when the expansion isView Solution
- 10View SolutionIn the case of diatomic gas, the heat given at constant pressure is that part of energy which is used for the expansion of gas, is
