Two gases are said to be in thermal equilibrium when they have same
Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1The heat capacity of one mole an ideal is found to be $C_V=3 R(1+a R T) / 2$, where $a$ is a constant. The equation obeyed by this gas during a reversible adiabatic expansion isView Solution
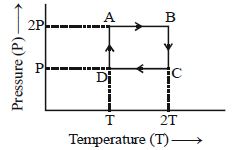
- 2One mole of an ideal gas having initial volume $V$, pressure $2P$ and temperature $T$ undergoes a cyclic process $ABCDA$ as shown below : The net work done in the complete cycle isView Solution
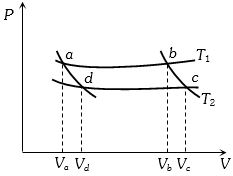
- 3In the following $P-V$ diagram two adiabatics cut two isothermals at temperatures $T_1$ and $T_2$ (fig.). The value of $\frac{{{V_a}}}{{{V_d}}}$ will beView Solution
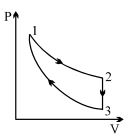
- 4Three processes form a thermodynamic cycle as shown on $P-V$ diagram for an ideal gas. Process $1 \rightarrow 2$ takes place at constant temperature $(300K$). Process $2 \rightarrow 3$ takes place at constant volume. During this process $40J$ of heat leaves the system. Process $3 \rightarrow 1$ is adiabatic and temperature $T_3$ is $275K$. Work done by the gas during the process $3 \rightarrow 1$ is ..... $J$View Solution
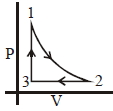
- 5Which of the following is an equivalent cyclic process corresponding to the thermodynamic cyclic given in the figure? where, $1 \rightarrow 2$ is adiabatic.View Solution
(Graphs are schematic and are not to scale)
- 6Carnot heat engine works with an ideal diatomic gas and an adiabatic volume expansion ratio $32$ . Then its efficiency is ....... $\%$View Solution
- 7A certain mass of gas at $273 K$ is expanded to $81$ times its volume under adiabatic condition. If $\gamma = 1.25$ for the gas, then its final temperature is ..... $^oC$View Solution
- 8The pressure in the tyre of a car is four times the atmospheric pressure at $300 K$. If this tyre suddenly bursts, its new temperature will be $(\gamma = 1.4)$View Solution
- 9A gas expands with temperature according to the relation $V = k{T^{2/3}}.$ What is the work done when the temperature changes by ${30^o}C$View Solution
- 10Two kg of water is converted into steam by boiling at atmospheric pressure. The volume changes from $2 \times {10^{ - 3}}\,{m^3}$ to $3.34{m^3}.$ The work done by the system is about ....... $kJ$View Solution
