A tyre filled with air $({27^o}C,$ and $2$ atm) bursts, then what is temperature of air ....... $^oC$ $(\gamma = 1.5)$
Medium
(a) $\frac{{{T_2}}}{{{T_1}}} = {\left( {\frac{{{P_2}}}{{{P_1}}}} \right)^{\frac{{\gamma - 1}}{\gamma }}}$==> $\frac{{{T_2}}}{{(273 + 27)}} = {\left( {\frac{1}{2}} \right)^{\frac{{1.5 - 1}}{{1.5}}}} = $${\left( {\frac{1}{2}} \right)^{\frac{1}{3}}} = \frac{1}{{2.5}}$
==> ${T_2} = \frac{{{T_1}}}{{1.25}} = \frac{{(273 + 27)}}{{1.25}} = 238\,K = - \,34.8^\circ C$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
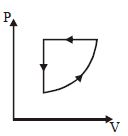
- 1For one complete cycle of a thermodynamic processes on a gas as shown in $P-V$ diagram. Which is trueView Solution
- 2An engine takes in $5$ moles of air at $20\,^{\circ} C$ and $1$ $atm,$ and compresses it adiabaticaly to $1 / 10^{\text {th }}$ of the original volume. Assuming air to be a diatomic ideal gas made up of rigid molecules, the change in its internal energy during this process comes out to be $X\, kJ$. The value of $X$ to the nearest integer isView Solution
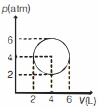
- 3An ideal gas undergoes a circular cycle centred at $4 \,atm , 4 L$ as shown in the diagram. The maximum temperature attained in this process is close toView Solution
- 4When heat energy of $1500\; Joules$, is supplied to a gas at constant pressure $2.1 \times {10^5}\;N/{m^2}$, there was an increase in its volume equal to $2.5 \times {10^{ - 3}}\;{m^3}$. The increase in internal energy of the gas in Joules is ...... $J$View Solution
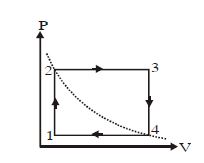
- 5One mole of an ideal gas undergoes a cyclic process, consisting of two isochores and two isobars. Temperature at $1$ and $3$ equal to $T_1$ and $T_3$ respectively. The work done by the gas over the cycle, if the point $2$ and $4$ lie on the same isothermView Solution
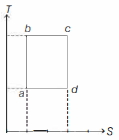
- 6An ideal gas is taken reversibly around the cycle $a-b-c-d-a$ as shown on the temperature $T$ - entropy $S$ diagram. The most appropriate representation of above cycle on a internal energy $U$ - volume $V$ diagram isView Solution
- 7$1 \,\,kg$ of a gas does $20\,\, kJ$ of work and receives $16 \,\,kJ$ of heat when it is expanded between two states. $A$ second kind of expansion can be found between the initial and final state which requires a heat input of $9\,\, kJ$. The work done by the gas in the second expansion is ....... $kJ$View Solution
- 8View SolutionPressure versus temperature graph of an ideal gas is shown in figure
- 9A polyatomic gas with six degrees of freedom does $25\,\,J$ of work when it is expanded at constant pressure. The heat given to the gas is ..... $J$View Solution
- 10In case of an adiabatic process the correct relation in terms of pressure $p$ and density $\rho $ of a gas isView Solution
