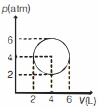
From given cyclic process,
Process equation is
$(p-4)^2+(V-4)^2=4 \quad \dots(i)$
Now, from $p V=n R T$
We can say that $T$ is maximum when $p V$ is maximum.
Now, for given cyclic process, $p V$ maximum occur when $p^2 V^2$ is maximum.
Now,
$p^2 V^2=p^2\left(4-(p-4)^2\right)$ [from Eq. $(i)$]
Now, $p^2 V^2$ is maximum when $\frac{d}{d p} p^2 V^2=0$
$\Rightarrow \quad \frac{d}{d p}\left(p^2 \cdot\left(p^2 \cdot\left(4-(p-4)^2\right)=0\right.\right.$
$\Rightarrow \quad p^2-8 p+14=0$
$=4 \pm \sqrt{2}$
and from Eq. $(i)$, we get
$p=4 \pm \sqrt{2}, V=4 \pm \sqrt{2}$
Taking positive values, we have
$(p V)_{\max } \Rightarrow p=4+\sqrt{2}$
and $V=4+\sqrt{2}$
So, by gas equation, we have
$T_{\max } =\frac{(p V)_{\max }}{R} \quad \text { [for } 1 \,mol \text { of gas] }$
$=\frac{(4+\sqrt{2})(4+\sqrt{2})}{R}$
$=\frac{16+2+2 \times 4 \times \sqrt{2}}{R}$
$=\frac{29.32}{R}=\frac{30}{R}$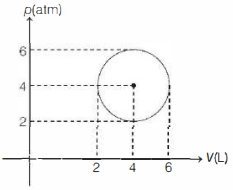
Download our appand get started for free
Similar Questions
- 1A thermodynamic cycle takes in heat energy at a high temperature and rejects energy at a lower temperature. If the amount of energy rejected at the low temperature is $3$ times the amount of work done by the cycle, the efficiency of the cycle isView Solution
- 2$Assertion :$ In isothermal process whole of the heat supplied to the body is converted into internal energy.View Solution
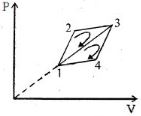
$Reason :$ According to the first law of thermodynamics : $\Delta Q = \Delta U + p\Delta V$ - 3The efficiency of a thermodynamic cycle $1-2-3- 1$ (see picture) is $20\%$ and for another thermodynamic cycle $1 - 3-4 - 1$ efficiency is equal to $10\%$. Determine the efficiency $\eta $ (in $\%$) of the thermodynamic cycle $1-2-3-4- 1.$The gas is assumed to be idealView Solution
- 4A thermodynamic system is taken form an initial state $i$ with internal energy $U_1=100 \ J$ to the final state along two different paths iaf and ibf, as schematically shown in the fire. The work done by the system along the paths $af$, ib and bf are $W _{ af }=200 \ J , W _{ ID }=50 \ J$ and $W _{ br }=100 \ J$ respectively. The heat supplied to the system along the path iaf, ib and bf are $Q_{\mid a t l} Q_{b r}$ and $Q_{10}$ respectively. If the internal energy of the sytem in the state $b$ is $U_b=$ $200 \ J$ and $Q_{l a t}=500 \ J$, the ratio $Q_{b J} / Q_{10}$ is:View Solution
- 5When an ideal gas $(\gamma = 5/3$) is heated under constant pressure, then what percentage of given heat energy will be utilised in doing external workView Solution
- 6View SolutionWhen a gas expands adiabatically
- 7The latent heat of vaporisation of water is $2240\, J/gm$. If the work done in the process of expansion of $1 \,g$ is $168 \,J$, then increase in internal energy is ....... $J$View Solution
- 8An ideal gas is expanded adiabatically at an initial temperature of $300 K$ so that its volume is doubled. The final temperature of the hydrogen gas is $(\gamma = 1.40)$View Solution
- 9For an ideal heat engine, the temperature of the source is $127\,^{\circ} C$. In order to have $60\, \%$ efficiency the temperature of the sink should be $........\,{ }^{\circ} C$. (Round off to the Nearest Integer)View Solution
- 10View SolutionWhat is the nature of change in internal energy in the following three thermodynamical processes shown in figure