According to law of equipartition of energy the molar specific heat of a diatomic gas at constant volume where the molecule has one additional vibrational mode is :-
JEE MAIN 2023, Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A container is filled with $20$ moles of an ideal diatomic gas at absolute temperature $T$. When heat is supplied to gas temperature remains constant but $8$ moles dissociate into atoms. Heat energy given to gas is .........View Solution
- 2An ideal gas is initially at temperature $T$ and volume $V.$ Its volume is increased by $\Delta V$ due to an increase in temperature $\Delta T,$ pressure remaining constant. The quantity $\delta = \Delta V/(V\Delta T)$ varies with temperature asView Solution
- 3One kg of a diatomic gas is at a pressure of $8 × 10^4\ N/m^2$. The density of the gas is $4\ kg/m^3$. What is the energy of the gas due to its thermal motion ?View Solution
- 4At which of the following temperature would the molecules of a gas have twice the average kinetic energy they have at $20°C$View Solution
- 5View SolutionA gas is filled in the cylinder shown in the figure. The two pistons are joined by a string. If the gas is heated, the pistons will
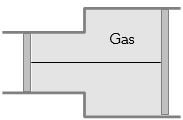
- 6If the pressure of an ideal gas contained in a closed vessel is increased by $0.5\%,$ the increase in temperature is $2K.$ The initial temperature of the gas is ...... $^oC$View Solution
- 7If the volume of the gas containing $n$ number of molecules is $V,$ then the pressure will decrease due to force of intermolecular attraction in the proportionView Solution
- 8A narrow glass tube, $80 \,cm$ long and opens at both ends, is half immersed in mercury, now the top of the tube is closed and is taken out of mercury. A column of mercury $20 \,cm$ long remains in the tube. Find atmospheric pressureView Solution
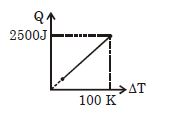
- 9One mole of a gas mixture is heated under constant pressure, and heat supplied $Q$ is plotted against temperature difference acquired. Find the approximate value of $\gamma $ for mixtureView Solution
- 10If $\mathrm{n}$ is the number density and $\mathrm{d}$ is the diameter of the molecule, then the average distance covered by a molecule between two successive collisions (i.e. mean free path) is represented by :View Solution
