An air bubble of volume $1.0\; cm ^{3}$ rises from the bottom of a lake $40\; m$ deep at a temperature of $12\,^{\circ} C$. To what volume (in $cm^3$) does it grow when it reaches the surface, which is at a temperature of $35\,^{\circ} C ?$
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionWhich of the following plots represents schematically the dependence of the time period of a pendulum, if measured and plotted as a function of the amplitude of its oscillations? (Note amplitude need not be small)
- 2The volume occupied by the molecules contained in $4.5\,kg$ water at $STP$, if the intermolecular forces vanish away is ........ $m ^{3}$View Solution
- 3View SolutionMolecular motion shows itself as
- 4Two non-reactive monoatomic ideal gases have their atomic masses in the ratio $2: 3$. The ratio of their partial pressures, when enclosed in a vessel kept at a constant temperature, is $4: 3$. The ratio of their densities is:View Solution
- 5The average degree of freedom per molecule of a gas is $6.$ The gas performs $25 \,J$ work, while expanding at constant pressure. The heat absorbed by the gas is ...... $J$View Solution
- 6The temperature at which the kinetic energy of oxygen molecules becomes double than its value at $27^{\circ}\,C$ is $............^{\circ}\,C$View Solution
- 7For two different gases $X$ and $Y,$ having degrees of freedom $f_1$ and $f_2$ and molar heat capacities at constant volume $C_{v_1}$ and $C_{v_2}$ respectively, for adiabatic process , the $\ln P$ versus $\ln V$ graph is plotted as shown :-View Solution
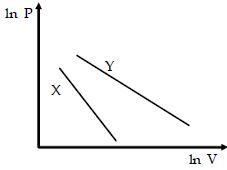
- 8Two identical adiabatic vessels are filled with oxygen at pressure $P_1$ and $P_2 (P_1 > P_2).$ The vessels are interconnected with each other by a nonconducting pipe. If $U_{01}$ and $U_{02}$ denote initial internal energy of oxygen in first and second vessel respectively and $U_{f_1}$ and $U_{f_2}$ denote final internal energy values, than :View Solution
- 9For a gas, the $r.m.s.$ speed at $800\, K$ isView Solution
- 10A soap bubble of radius $r$ has monoatomic ideal- gas inside. The gas is heated in such a manner that bubble remains in mechanical equillibrium. Assuming that the soap material of the bubble has no heat capacity, the molar heat capacity of the gas in the process will beView Solution
(Neglect atmospheric pressure)
