Two identical adiabatic vessels are filled with oxygen at pressure $P_1$ and $P_2 (P_1 > P_2).$ The vessels are interconnected with each other by a nonconducting pipe. If $U_{01}$ and $U_{02}$ denote initial internal energy of oxygen in first and second vessel respectively and $U_{f_1}$ and $U_{f_2}$ denote final internal energy values, than :
Diffcult
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1$2\, gm$ of ${O_2}$ gas is taken at $27°C$ and pressure $76\, cm.$ Of $Hg$. Then find out volume of gas (in litre)View Solution
- 2View SolutionThe root mean square speed of the molecules of a gas is
- 3A diatomic gas follows equation $PV^m =$ constant, during a process. What should be the value of $m$ such that its molar heat capacity during process $= R$View Solution
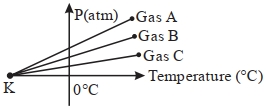
- 4For three low density gases $A, B, C$ pressure versus temperature graphs are plotted while keeping them at constant volume, as shown in the figure.View Solution
The temperature corresponding to the point ' $K$ ' is $...........\,{}^{\circ}\,C$
- 5The average degree of freedom per molecule of a gas is $6$. The gas performs $25\ J$ work, while expanding at constant pressure. The heat absorbed by the gas is .... $J$View Solution
- 6View SolutionSelect the incorrect statement about the specific heats of a gaseous system.
- 7$Assertion :$ Mean free path of a gas molecules varies inversely as density of the gas.View Solution
$Reason :$ Mean free path varies inversely as pressure of the gas. - 8The number of molecules in one litre of an ideal gas at $300 \,{K}$ and $2$ atmospheric pressure with mean kinetic energy $2 \times 10^{-9}\, {J}$ per molecules is $....\, \times 10^{11}$View Solution
- 9The mean free path of molecules of a certain gas at $STP$ is $1500\,d$, where $d$ is the diameter of the gas molecules. While maintaining the standard pressure, the mean free path of the molecules at $373\,K$ is approximately $..........\,d$View Solution
- 10The temperature of the hydrogen at which the average speed of its molecules is equal to that of oxygen molecules at a temperature of $31\,^oC,$ is ........ $^oC$View Solution
