An electric kettle takes $4\, A$ current at $220\,V.$ How much time will it take to boil $1\,\, kg$ of water circuits are consequences of from temperature $20\,^o C\, ?$ The temperature of $(a)$ conservation of energy and electric charge boiling water is $100\,^o C.$ ............... $min$
AIPMT 2008, Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
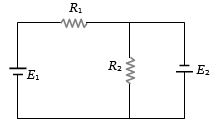
- 1Two resistances ${R_1}$ and ${R_2}$ are joined as shown in the figure to two batteries of $e.m.f.$ ${E_1}$ and ${E_2}$. If ${E_2}$ is short-circuited, the current through ${R_1}$ isView Solution
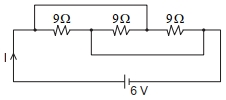
- 2The current $I$ flowing through the given circuit will be $.....A$.View Solution
- 3View SolutionAssertion : Long distance power transmission is done at high voltage.
Reason : At high voltage supply power losses are less.
- 4The effective resistance of the network between points $A$ and $B$ isView Solution
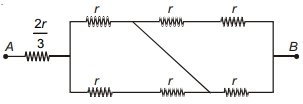
- 5Two resistors of resistance, $100\,\Omega$ and $200\,\Omega$ are connected in parallel in an electrical circuit. The ratio of the thermal energy developed in $100\,\Omega$ to that in $200\,\Omega$ in a given time is:View Solution
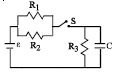
- 6The circuit shown in the figure consists of a battery of $emf$ $\varepsilon = 10 \,V$ ; a capacitor of capacitance $C = 1.0$ $ \mu F$ and three resistor of values $R_1 = 2$ $\Omega$ , $R_2 = 2$ $\Omega$ and $R_3 = 1$ $\Omega$ . Initially the capacitor is completely uncharged and the switch $S$ is open. The switch $S$ is closed at $t = 0.$View Solution
- 7The thermo $e.m.f.$ of a thermo-couple is $25\,\mu V{/^o}C$ at room temperature. A galvanometer of $40\, ohm$ resistance, capable of detecting current as low as ${10^{ - 5}}\,A,$ is connected with the thermocouple. The smallest temperature difference that can be detected by this system is ................ $^oC$View Solution
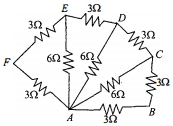
- 8Six resistors of $3 \;\Omega$ each are connected along the sides of a hexagon and three resistors of $6\; \Omega$ each are connected along $A C, A D$ and $A E$ as shown in the figure. The equivalent resistance between $A$ and $B$ is equal toView Solution
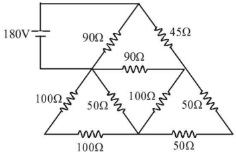
- 9In the circuit shown in the figure, find the current in $45\, \Omega$ $..........\,A$View Solution
- 10The resistance of $10\, metre$ long potentiometer wire is $1\,ohm/meter$. A cell of $e.m.f.$ $2.2\, volts$ and a high resistance box are connected in series to this wire. The value of resistance taken from resistance box for getting potential gradient of $2.2\, millivolt/metre$ will be ............... $\Omega $View Solution
