The number of molecules in a gas at pressure $1.64 \times {10^{ - 3}}$atmospheres and temperature $200\, K$ having the volume $1 \,cc$ are
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1The expansion of an ideal gas of mass $m$ at a constant pressure $P$ is given by the straight line $B$. Then the expansion of the same ideal gas of mass $2 \,\,m$ at a pressure $2P$ is given by the straight lineView Solution
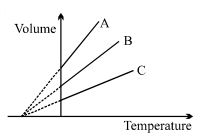
- 2View SolutionIf the mean free path of atoms is doubled then the pressure of gas will become
- 3View SolutionSelect the incorrect statement about Maxwell's speed distribution
- 4The volume of a gas at pressure $21 \times {10^4}\,N/{m^2}$ and temperature $27^o C$ is $83 $ litres. If $R = 8.3\ J/mol/K$, then the quantity of gas in $gm-mole$ will beView Solution
- 5At what temperature will the oxygen molecules have the same root mean square speed as hydrogen molecules at $200 \,K$ ....... $K$View Solution
- 6In Maxwell's speed distribution curve, for $N_2$ gas, the average of $\mid$ relative velocity (in $m/s$) $\mid$ between two molecules at $300 \,K$ will beView Solution
- 7A monatomic ideal gas undergoes a process in which the ratio of $\mathrm{P}$ to $\mathrm{V}$ at any instant is constant and equals to $1$. What is the molar heat capacity of the gas?View Solution
- 8View SolutionThe kinetic energy per gm mol for a diatomic gas at room temperature is
- 9View SolutionAccording to kinetic theory of gases,
- 10The volume $V$ of an enclosure contains a mixture of three gases, $16\, g$ of oxygen, $28\, g$ of nitrogen and $44\, g$ of carbon dioxide at absolute temperature $T$. Consider $R$ as universal gas constant. The pressure of the mixture of gases isView Solution
