An engine is supposed to operate between two reservoirs at temperature $727°C$ and $227°C.$ The maximum possible efficiency of such an engine is
Medium
(a) $\eta = \frac{{{T_1} - {T_2}}}{{{T_1}}} = \frac{{(273 + 727) - (273 + 227)}}{{273 + 727}}$$ = \frac{{1000 - 500}}{{1000}} = \frac{1}{2}$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1Carnot cycle is plotted in $P-V$ graph. Which portion represents an isothermal expansion?View Solution
- 2View SolutionIn the adiabatic compression, the decrease in volume is associated with
- 3An ideal gas heat engine operates in a Carnot cycle between $227^o C$ and $127^o C$. It absorbs $6\,kcal$ at the higher temperature. The amount of heat (in $kcal$) converted into work is equal toView Solution
- 4An engineer claims to have made an engine delivering $10 kW$ power with fuel consumption of $1\,g\,{s^{ - 1}}$. The calorific value of fuel is $2k cal/g$. His claimView Solution
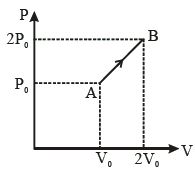
- 5An ideal gas follows a process described by $p V^2=C$ from $\left(p_1, V_1, T_1\right)$ to $\left(p_2, V_2, T_2\right)$ and $C$ is a constant. Then,View Solution
- 6One mole of helium is adiabatically expanded from its initial state $({P_i},{V_i},{T_i})$ to its final state $({P_f},{V_f},{T_f})$. The decrease in the internal energy associated with this expansion is equal toView Solution
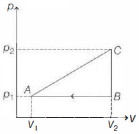
- 7An ideal gas with constant heat capacity $C_V=\frac{3}{2} n R$ is made to carry out a cycle that is depicted by a triangle in the figure given below. The following statement is true about the cycle.View Solution
- 8A monoatomic gas is taken through a process $TP^{-1/3} =$ constant. If heat is given to the gasView Solution
- 9$Assertion :$ The heat supplied to a system is always equal to the increase in its internal energy.View Solution
$Reason :$ When a system changes from one thermal equilibrium to another, some heat is absorbed by it. - 10The $P-V$ diagram of $2\,g$ of helium gas for a certain process $A$ $\to$ $B$ is shown in the figure. What is the heat given to the gas during the process $A$ $\to$ $B$?View Solution