An ideal gas heat engine operates in a Carnot cycle between $227^o C$ and $127^o C$. It absorbs $6\,kcal$ at the higher temperature. The amount of heat (in $kcal$) converted into work is equal to
AIPMT 2003, Medium
(c) Efficiency of a carnot engine is given by $\eta = 1 - \frac{{{T_2}}}{{{T_1}}}$
or $\frac{W}{Q} = 1 - \frac{{{T_2}}}{{{T_1}}}$==> $\frac{W}{6} = 1 - \frac{{(273 + 127)}}{{(273 + 227)}}$==> $W = 1.2\,k\,cal$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1Consider $1 \,kg$ of liquid water undergoing change in phase to water vapour at $100^{\circ} C$. At $100^{\circ} C$, the vapour pressure is $1.01 \times 10^5 \,N - m ^2$ and the latent heat of vaporization is $22.6 \times 10^5 \,Jkg ^{-1}$. The density of liquid water is $10^3 \,kg m ^{-3}$ and that of vapour is $\frac{1}{1.8} \,kg m ^{-3}$. The change in internal energy in this phase change is nearly ............ $\,J kg ^{-1}$View Solution
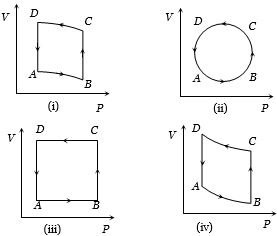
- 2In the diagrams $(i)$ to $(iv)$ of variation of volume with changing pressure is shown. A gas is taken along the path $ABCD. $ The change in internal energy of the gas will beView Solution
- 3An ideal gas is expanding such that $\mathrm{PT}^2=$ constant. The coefficient of volume expansion of the gas isView Solution
- 4View SolutionThe efficiency of a Carnot engine depends upon
- 5View SolutionIn thermodynamics, heat and work are
- 6A diatomic gas $(\gamma=1.4)$ does $400 J$ of work when it is expanded isobarically. The heat given to the gas in the process is ............ $J$View Solution
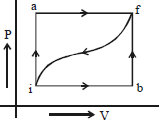
- 7When a system is taken from a state $i$ to $f$ along the path $iaf$ (as shown in the figure). $Q = 50\, cal$ and $W = 20\, cal$ ; along path $ibf,$ $Q = 36\, cal.$View Solution
$(i)$ What is $W$ along path $ibf$ ?
$(ii)$ If $W = 13$ cal for path $fi$, what is $Q$ for the path $fi$ ?
$(iii)$ Take $E_{int,i} = 10\,\, cal$ then what is $E_{int,f}$ ?
- 8A heat engine operates between a cold reservoir at temperature ${T}_{2}=400\, {K}$ and a hot reservoir at temperature ${T}_{1} .$ It takes $300 \,{J}$ of heat from the hot reservoir and delivers $240\, {J}$ of heat to the cold reservoir in a cycle. The minimum temperature of the hot reservoir has to be $....{K}$View Solution
- 9View SolutionWhich of the following statements is correct for any thermodynamic system
- 10The first law of thermodynamics can be written as $ \Delta U = \Delta Q + \Delta W$ for an ideal gas. Which of the following statements is correct?View Solution
