Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
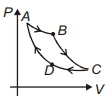
- 1An ideal gas undergoes the process $1 \rightarrow 2$ as shown in the figure, the heat supplied and work done in the process is $\Delta \,\,Q$ and $\Delta \,\,W$ respectively. The ratio $\Delta \,\,Q :$ $\Delta \,\,W$ isView Solution
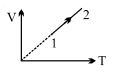
- 2One mole of an ideal monoatomic gas undergoes the following four reversible processes:View Solution
Step $1$ It is first compressed adiabatically from volume $8.0 \,m ^{3}$ to $1.0 \,m ^{3}$.
Step $2$ Then expanded isothermally at temperature $T_{1}$ to volume $10.0 \,m ^{3}$.
Step $3$ Then expanded adiabatically to volume $80.0 \,m ^{3}$.
Step $4$ Then compressed isothermally at temperature $T_{2}$ to volume $8.0 \,m ^{3}$.
Then, $T_{1} / T_{2}$ is
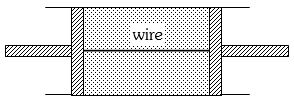
- 3A cylindrical tube of uniform cross-sectional area $A$ is fitted with two air tight frictionless pistons. The pistons are connected to each other by a metallic wire. Initially the pressure of the gas is $P_0$ and temperature is $T_0$, atmospheric pressure is also $P_0$. Now the temperature of the gas is increased to $2T_0$, the tension in the wire will beView Solution
- 4Starting with the same initial conditions, an ideal gas expands from volume $V_{1}$ to $V_{2}$ in three different ways. The work done by the gas is $W_{1}$ if the process is purely isothermal. $W _{2}$. if the process is purely adiabatic and $W _{3}$ if the process is purely isobaric. Then, choose the coned optionView Solution
- 5A system is provided with $200 \,cal$ of heat and the work done by the system on the surrounding is $40 \,J$. Then its internal energyView Solution
- 6$200\,g$ water is heated from $40\,^oC$ to $60\,^oC.$ Ignoring the slight expansion of water, the change in its internal energy is close to ...... $kJ$ (Given specific heat of water $=4184\,J/kgK$ )View Solution
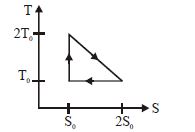
- 7View SolutionThe temperature-entropy diagram of a reversible engine cycle is given in the figure. Its efficiency is
- 8$P_i, V_i$ and $P_f$, $V_f$ are initial and final pressures and volumes of a gas in a thermodynamic process respectively. If $PV^n =$ constant, then the amount of work done isView Solution
- 9Column $I$ Contains a list of processes involving expansion of an ideal gas. Match this with Column $II$ describing the thermodynamic change during this process. Indicate your answer by darkening the appropriate bubbles of the $4 \times 4$ matrix given in the $ORS$.View Solution
Column $I$ Column $II$ $(A)$ An insulated container has two chambers separated by a valve. Chamber $I$ contains an ideal gas and the Chamber $II$ has vacuum. The valve is opened. $(p)$ The temperature of the gas decreases $(B)$ An ideal monoatomic gas expands to twice its original volume such that its pressure $\mathrm{P} \propto \frac{1}{\mathrm{~V}^2}$, where $\mathrm{V}$ is the volume of the gas $(q)$ The temperature of the gas increases or remains constant $(C)$ An ideal monoatomic gas expands to twice its original volume such that its pressure $\mathrm{P} \propto \frac{1}{\mathrm{~V}^{4 / 3}}$, where $\mathrm{V}$ is its volume $(r)$ The gas loses heat $(D)$ An ideal monoatomic gas expands such that its pressure $\mathrm{P}$ and volume $\mathrm{V}$ follows the behaviour shown in the graph $Image$ $(s)$ The gas gains heat 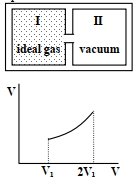
- 10$N _{2}$ gas is heated from $300\, K$ temperature to $600\, K$ through an isobaric process. Then find the change in entropy of the gas. $( n =1 mole )$ (in $J/K$)View Solution