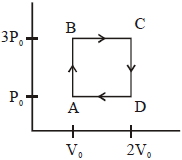
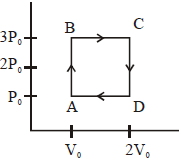
An engine operates by taking a monatomic ideal gas through the cycle shown in the figure. The percentage efficiency of the engine is close to $.......\%$

JEE MAIN 2020, Diffcult
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1An ideal system can be brought from state $A$ to $B$ through four paths as shown in the figure. The energy given to the system is minimum inView Solution
- 2View SolutionIn which thermodynamic process, volume remains same
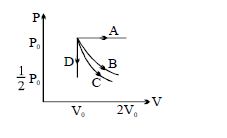
- 3The $PV$ diagram shows four different possible reversible processes performed on a monatomic ideal gas. Process $A$ is isobaric (constant pressure). Process $B$ is isothermal (constant temperature). Process $C$ is adiabatic. Process $D$ is isochoric (constant volume). For which process$(es)$ does the temperature of the gas decrease?View Solution
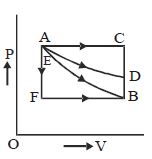
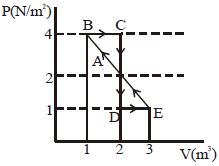
- 4One mole of a monoatomic gas is carried along process $ABCDEA$ as shown in diagram. Find the net work done by gas ......... $J$View Solution
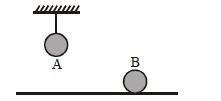
- 5Two identical balls, $A$ and $B$ , of uniform composition and initially at the same temperature, each absorb exactly the same amount of heat. $A$ is hanging down from the ceiling while $B$ rests on the horizontal floor in the same room. Assuming no subsequent heat loss by the balls, which of the following statements is correct about their final temperatures, $T_A$ and $T_B$ , once the balls have reached their final state?View Solution
- 6The heat energy required to raise the temperature of $5\,moles$ of an ideal gas to $5\,K$ at constant pressure is $600\,J$ . How much heat (in $J$ ) is required to raise the same mass of the same gas to $5\,K$ at constant volume ? (Take $R = 8.3\,J/mole-^oK$ )View Solution
- 7View SolutionA measure of the degree of disorder of a system is known as
- 8A van der Waal's gas obeys the equation of state $\left(p+\frac{n^2 a}{V^2}\right)(V-n b)=n R T$. Its internal energy is given by $U=C T-\frac{n^2 a}{V}$. The equation of a quasistatic adiabat for this gas is given byView Solution
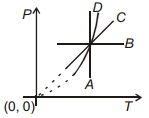
- 9Following figure shows $P-T$ graph for four processes $A, B, C$ and $D$. Select the correct alternative.View Solution
- 10An ideal gas undergoes the process $1 \rightarrow 2$ as shown in the figure, the heat supplied and work done in the process is $\Delta \,\,Q$ and $\Delta \,\,W$ respectively. The ratio $\Delta \,\,Q :$ $\Delta \,\,W$ isView Solution