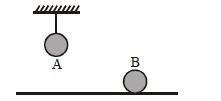
Two identical balls, $A$ and $B$ , of uniform composition and initially at the same temperature, each absorb exactly the same amount of heat. $A$ is hanging down from the ceiling while $B$ rests on the horizontal floor in the same room. Assuming no subsequent heat loss by the balls, which of the following statements is correct about their final temperatures, $T_A$ and $T_B$ , once the balls have reached their final state?

Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A gas undergoes a change of state during which $100 J$ of heat is supplied to it and it does $20 J$ of work. The system is brought back to its original state through a process during which $20 J$ of heat is released by the gas. The work done by the gas in the second process is ....... $J$View Solution
- 2The graph of pressure $(P)$ and volume $(V)$ according to $PV^n = C$, here $n = 1.4$View Solution
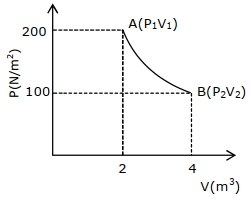
- 3One mole of an ideal gas at $27^{\circ} {C}$ is taken from ${A}$ to ${B}$ as shown in the given ${PV}$ indicator diagram. The work done by the system will be $......\times 10^{-1} \,{J}$View Solution
[Given : $R=8.3\, {J} /\,mole\,{K}, \ln 2=0.6931$ ] (Round off to the nearest integer)
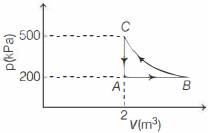
- 4A constant amount of an ideal gas undergoes the cyclic process $A B C A$ in the $p-V$ graph shown below. The path $B C$ is an isothermal. The work done by the gas during one complete cycle, beginning and ending at $A$ is nearly .......... $\,kJ$View Solution
- 5In a mechanical refrigerator, the low temperature coils are at a temperature of $-23°C$ and the compressed gas in the condenser has a temperature of $27°C.$ The theoretical coefficient of performance isView Solution
- 6A Car not engine whose low temperate reservoir is at $7\,^oC$ has an efficiency of $50\%$ . It is desired to increase the efficiency to $70\%$ . By how many degrees should the temperature of the high temperature reservoir be increased .... $K$View Solution
- 7Diatomic gas is used in carnot heat engine. If efficiency of given carnot heat engine is $80\%$ , then find the ratio of initial volume to final volume of gas during adiabatic expansionView Solution
- 8$Assertion :$ The isothermal curves intersect each other at a certain point.View Solution
$Reason :$ The isothermal change takes place slowly, so, the isothermal curves have very little slope. - 9Let $\eta_{1}$ is the efficiency of an engine at $T _{1}=447^{\circ}\,C$ and $T _{2}=147^{\circ}\,C$ while $\eta_{2}$ is the efficiency at $T _{1}=947^{\circ}\,C$ and $T _{2}=47^{\circ}\,C$. The ratio $\frac{\eta_{1}}{\eta_{2}}$ will be.View Solution
- 10View Solution“Heat cannot by itself flow from a body at lower temperature to a body at higher temperature” is a statement or consequence of
