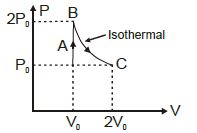
An ideal gas expands from volume $V_1$ to $V_2$. This may be achieved by either of the three processes: isobaric, isothermal and adiabatic. Let $\Delta U$ be the change in internal energy of the gas, $Q$ be the quantity of heat added to the system and $W$ be the work done by the system on the gas. Identify which of the following statements is false for $\Delta U$?
Advanced
We know that, $\Delta U \propto \Delta T$
For isotherm, $\Delta T=0 \Rightarrow \Delta U=0$
For adiabat, $\Delta Q=0 \Rightarrow \Delta U=-\Delta W<0$
For isobar, $\Delta V \propto \Delta T \Rightarrow \Delta T>0 \Rightarrow \Delta U>0$
Let the internal energy change be $\Delta U_{1}, \Delta U_{2}, \Delta U_{3}$ for the isotherm, adiabat, isobar respectively.
Then $\Delta U_{3}>\Delta U_{1}>\Delta U_{2}$
Thus option $b$ is only false
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A monoatomic ideal gas, initially at temperature ${T_1},$ is enclosed in a cylinder fitted with a frictionless piston. The gas is allowed to expand adiabatically to a temperature. ${T_2}$ by releasing the piston suddenly. If ${L_1}$ and ${L_2}$ are the lengths of the gas column before and after expansion respectively, then ${T_1}/{T_2}$ is given byView Solution
- 2An ideal gas is subjected to an isothermal expansion such that its volume changes from $V_i$ to $V_f$ and pressure from $P_i$ to $P_f$. The work done on the gas is :View Solution
- 3View SolutionA gas is compressed adiabatically till its temperature is doubled. The ratio of its final volume to initial volume will be
- 4A gas expands with temperature according to the relation $V = k{T^{2/3}}.$ What is the work done when the temperature changes by ${30^o}C$View Solution
- 5Three moles of an ideal monoatomic gas perform a cycle as shown in the figure. The gas temperature in different states are: $T_1 = 400\, K, T_2 = 800\, K, T_3 = 2400\,K$ and $T_4 = 1200\,K.$ The work done by the gas during the cycle is ........ $kJ$View Solution
- 6$Assertion :$ The isothermal curves intersect each other at a certain point.View Solution
$Reason :$ The isothermal change takes place slowly, so, the isothermal curves have very little slope. - 7A Carnot freezer takes heat from water at $0\,^oC$ inside it and rejects it to the room at a temperature of $27\,^oC$. The latent heat of ice is $336 \times 10^3\, J\,kg^{-1}$. lf $5\, kg$ of water at $0\,^oC$ is converted into ice at $0\,^oC$ by the freezer, then the energy consumed by the freezer is close toView Solution
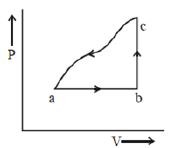
- 8A sample of an ideal gas is taken through the cyclic process $abca$ as shown in the figure. The change in the internal energy of the gas along the path $ca$ is $-180\, J$. The gas absorbs $250\, J$ of heat along the path $ab$ and $60\, J$ along the path $bc$. The work done by the gas along the path $abc$ is ..... $J$View Solution
- 9For an ideal heat engine, the temperature of the source is $127\,^{\circ} C$. In order to have $60\, \%$ efficiency the temperature of the sink should be $........\,{ }^{\circ} C$. (Round off to the Nearest Integer)View Solution
- 10A diatomic ideal gas undergoes a thermodynamic change according to the $P-V$ diagram shown in the figure. The heat given the gas in $AB$View Solution