An ideal gas is subjected to an isothermal expansion such that its volume changes from $V_i$ to $V_f$ and pressure from $P_i$ to $P_f$. The work done on the gas is :
AIIMS 2011, Easy
The work done in expansion of gas
$\mathrm{W}=\mathrm{nRT}\, \ell n \frac{\mathrm{V}_{\mathrm{f}}}{\mathrm{V}_{\mathrm{i}}}=\mathrm{nRT}\, \ell n \frac{\mathrm{P}_{1}}{\mathrm{P}_{\mathrm{f}}}$
The work done on the gas
$=-n R T \ell n\left(\frac{P_{f}}{P_{1}}\right)$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1An ideal monoatomic gas with pressure $P$, volume $V$ and temperature $T$ is expanded isothermally to a volume $2\, V$ and a final pressure $P_i$. If the same gas is expanded adiabatically to a volume $2\,V$, the final pressure is $P_a$ . The ratio $\frac{{{P_a}}}{{{P_i}}}$ isView Solution
- 2One mole of a perfect gas in a cylinder fitted with a piston has a pressure $P,$ volume $V$ and temperature $T.$ If the temperature is increased by $1 \,K$ keeping pressure constant, the increase in volume isView Solution
- 3A gas at $NTP$ is suddenly compressed to one-fourth of its original volume. If $\gamma $ is supposed to be $\frac{3}{2}$, then the final pressure is........ atmosphereView Solution
- 4Work done by a system under isothermal change from a volume ${V_1}$ to ${V_2}$ for a gas which obeys Vander Waal's equation $(V - \beta n)\,\left( {P + \frac{{\alpha {n^2}}}{V}} \right) = nRT$View Solution
- 5View SolutionWhich of the following laws of thermodynamics defines the term internal energy?
- 6A piece of hot copper at $100^{\circ} C$ is plunged into a pond at $30^{\circ} C$. The copper cools down to $30^{\circ} C$, while the pond being huge stays at its initial temperature. Then,View Solution
- 7View SolutionWork done in the cyclic process shown in figure is ...........
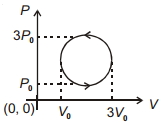
- 8A gas is compressed from a volume of $2\,m^3$ to a volume of $1\, m^3$ at a constant pressure of $100\, N/m^2$. Then it is heated at constant volume by supplying $150\, J$ of energy. As a result, the internal energy of the gasView Solution
- 9Work done in the given $P-V$ diagram in the cyclic process isView Solution
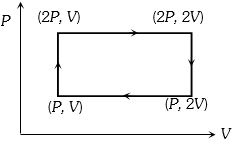
- 10One mole of an ideal gas expands at a constant temperature of $300 \,K$ from an initial volume of $10\, litres$ to a final volume of $20\, litres$. The work done in expanding the gas is ...... $J.$ $(R = 8.31 J/mole-K)$View Solution
