In an adiabatic process where in pressure is increased by $\frac{2}{3}\% $ if $\frac{{{C_p}}}{{{C_v}}} = \frac{3}{2},$ then the volume decreases by about
Medium
$\frac{\Delta \mathrm{P}}{\mathrm{P}}=-\gamma \frac{\Delta \mathrm{V}}{\mathrm{V}} \quad \frac{2}{3} \times \frac{1}{\gamma}=\frac{\Delta \mathrm{V}}{\mathrm{V}} \quad \frac{\Delta \mathrm{V}}{\mathrm{V}}=-\frac{4}{9} \%$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
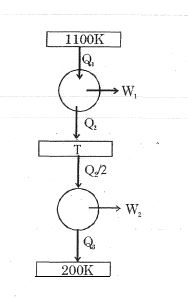
- 1Consider the given series combination of carnot cycles. If $W_1 = W_2$ then the value of $T$ is ...... $K$ (all temperatures are maintained at their respective values)View Solution
- 2Under isothermal condition, the pressure of a gas is given by $P = aV ^{-3}$, where $a$ is a constant and $V$ is the volume of the gas. The bulk modulus at constant temperature is equal to $..........\,P$View Solution
- 3An ideal gas expands in such a way that $PV^2 =$ constant throughout the process.View Solution
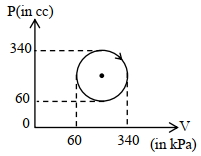
- 4View SolutionThe heat absorbed by a system in going through the given cyclic process is:
- 5A Carnot engine with sink's temperature at $17\,^oC$ has $50\%$ efficiency. By how much should its source temperature be changed to increases its efficiency to $60\%$ ?...... $K$View Solution
- 6View SolutionOne mole of a monoatomic gas and one mole of a diatomic gas are initially in the same state. Both gases are expanded isothermally and then adiabatically, such that they acquire the same final state. Choose the correct statement.
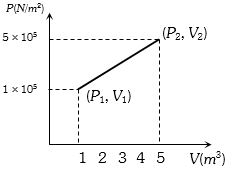
- 7A system changes from the state $({P_1},\;{V_1})$ to $({P_2},{V_2})$ as shown in the figure. What is the work done by the systemView Solution
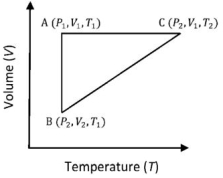
- 8A reversible cyclic process for an ideal gas is shown below. Here, $P, V$, and $T$ are pressure, volume and temperature, respectively. The thermodynamic parameters $q, w, H$ and $U$ are heat, work, enthalpy and internal energy, respectively.View Solution
(image)
The correct option ($s$) is (are)
$(A)$ $q_{A C}=\Delta U_{B C}$ and $W_{A B}=P_2\left(V_2-V_1\right)$
$(B)$ $\mathrm{W}_{\mathrm{BC}}=\mathrm{P}_2\left(\mathrm{~V}_2-\mathrm{V}_1\right)$ and $\mathrm{q}_{\mathrm{BC}}=\mathrm{H}_{\mathrm{AC}}$
$(C)$ $\Delta \mathrm{H}_{\mathrm{CA}}<\Delta \mathrm{U}_{\mathrm{CA}}$ and $\mathrm{q}_{\mathrm{AC}}=\Delta \mathrm{U}_{\mathrm{BC}}$
$(D)$ $\mathrm{q}_{\mathrm{BC}}=\Delta \mathrm{H}_{\mathrm{AC}}$ and $\Delta \mathrm{H}_{\mathrm{CA}}>\Delta \mathrm{U}_{\mathrm{CA}}$
- 9View SolutionFor a reversible process, necessary condition is
- 10View SolutionThe specific heat of a gas in an isothermal process is
