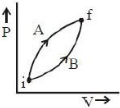
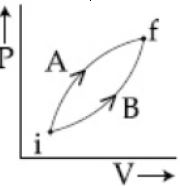
Following figure shows two processes $A$ and $B$ for a gas. If $\Delta Q_A$ and $\Delta Q_B$ are the amount of heat absorbed by the system in two cases, and $\Delta U_A$ and $\Delta U_B$ are changes in internal energies, respectively, then

JEE MAIN 2019, Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A motor-car tyre has a pressure of $2\, atm$ at $27\,^oC$. It suddenly burst's. If $\left( {\frac{{{C_p}}}{{{C_v}}}} \right) = 1.4$ for air, find the resulting temperatures (Given $4^{1/7} = 1.219$)View Solution
- 2The coefficient of performance of a Carnot refrigerator working between ${30^o}C$ and ${0^o}C$ isView Solution
- 3A gas expands adiabatically at constant pressure such that its temperature $T \propto \frac{1}{{\sqrt V }}$, the value of ${C_P}/{C_V}$ of gas isView Solution
- 4View SolutionIn thermodynamic processes which of the following statements is not true?
- 5A Carnot engine works between $27^{\circ} C$ and $127^{\circ} C$. Heat supplied by the source is $500\, J$. then heat ejected to the sink is (in $J$)View Solution
- 6Agas expands such that its initial and final temperature are equal. Also, the process followed by the gas traces a straight line on the $P-V$ diagram :View Solution
- 7$A$ reversible engine converts one-sixth of the heat input into work. When the temperature of the sink is reduced by $ 62^oC$, the efficiency of the engine is doubled. The temperatures of the source and sink areView Solution
- 8Consider the following volume-temperature $( V - T )$ diagram for the expansion of $5$ moles of an ideal monoatomic gas.View Solution
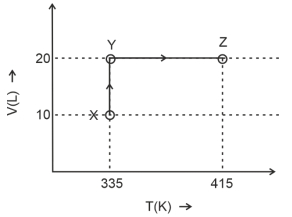
Considering only $P-V$ work is involved, the total change in enthalpy (in Joule) for the transformation of state in the sequence $X \rightarrow Y \rightarrow Z$ is $\qquad$
[Use the given data: Molar heat capacity of the gas for the given temperature range, $C _{ v , m }=12 J K ^{-1} mol ^{-1}$ and gas constant, $R =8.3 J K ^{-1} mol ^{-1}$ ]
- 9Two cylinders $A$ and $B$ fitted with pistons contain equal amounts of an ideal diatomic gas at $300 K$ . The piston of $A$ is free to move while that of $B$ is held fixed. The same amount of heat is given to the gas in each cylinder. If the rise in temperature of the gas in $A$ is $30 K$ , then the rise in temperature of the gas in $B$ is ..... $K$View Solution
- 10In a mechanical refrigerator, the low temperature coils are at a temperature of $-23°C$ and the compressed gas in the condenser has a temperature of $27°C.$ The theoretical coefficient of performance isView Solution