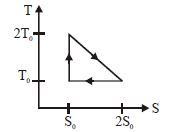
$Q=T . \Delta S$
According to $T-S$ diagram, the system absortbs heat $\mathrm{Q}_{1}$ from $\mathrm{A}$ to $\mathrm{B}$
$\mathrm{Q}_{1}=$ (Average temperature).$\Delta S$
$=\left(\frac{2 \mathrm{T}_{0}+\mathrm{T}_{0}}{2}\right)\left(2 \mathrm{S}_{0}-\mathrm{S}_{0}\right)$
$\mathrm{Q}_{1}=\frac{3}{2} \mathrm{T}_{0} \mathrm{S}_{0}$ $...(i)$
The system rejects heat $\mathrm{Q}_{2}$ from $\mathrm{B}$ to $\mathrm{C}$.
$\mathrm{Q}_{2}=\mathrm{T}_{0}\left(2 \mathrm{S}_{0}-\mathrm{S}_{0}\right)=\mathrm{T}_{0} \mathrm{S}_{0}$ $...(ii)$
From $\mathrm{C}$ to $\mathrm{A}$ entropy remains constant, hence The efficiency $\eta$ of the engine is therefore
$\eta=1-\frac{\mathrm{Q}_{2}}{\mathrm{Q}_{1}}=1-\frac{\mathrm{T}_{0} \mathrm{S}_{0}}{\frac{3}{2} \mathrm{T}_{0} \mathrm{S}_{0}}$
$=1-\frac{2}{3}=\frac{1}{3}$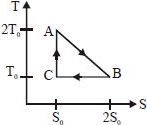
Download our appand get started for free
Similar Questions
- 1If the amount of heat given to a system be $35$ joules and the amount of work done by the system be $ - 15$ joules, then the change in the internal energy of the system is .... $joules$View Solution
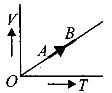
- 2The volume $( V)$ of a monatomic gas varies with its temperature $(T)$ , as shown in the graph. The ratio of work done by the gas , to the heat absorbed by it, when it undergoes a change from state $A$ to state $B$ , isView Solution
- 3The volume of an ideal gas is $1$ litre and its pressure is equal to $72cm$ of mercury column. The volume of gas is made $900\, cm^3$ by compressing it isothermally. The stress of the gas will be ...... $cm$ (mercury)View Solution
- 4A Carnot's engine working between $400\, K$ and $800\, K$ has a work output of $1200\, J$ per cycle. The amount of heat energy supplied to the engine from the source in each cycle is ........... $J$View Solution
- 5An engine has an efficiency of $0.25$ when temperature of sink is reduced by $58\,^oC$, if its efficiency is doubled, then the temperature of the source is ..... $^oC$View Solution
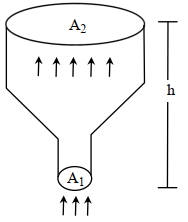
- 6An ideal gas of density $\rho=0.2 kg m ^{-3}$ enters a chimney of height $h$ at the rate of $\alpha=0.8 kg s ^{-1}$ from its lower end, and escapes through the upper end as shown in the figure. The cross-sectional area of the lower end is $A_1=0.1 m ^2$ and the upper end is $A_2=0.4 m ^2$. The pressure and the temperature of the gas at the lower end are $600 Pa$ and $300 K$, respectively, while its temperature at the upper end is $150 K$. The chimney is heat insulated so that the gas undergoes adiabatic expansion. Take $g=10 ms ^{-2}$ and the ratio of specific heats of the gas $\gamma=2$. Ignore atmospheric pressure.View Solution
Which of the following statement($s$) is(are) correct?
- 7A cyclic process $ABCD$ is shown in the figure $P-V$ diagram. Which of the following curves represent the same processView Solution
- 8Given below are two statements : one is labelled as Assertion A and the other is labelled as Reason $R$. Assertion A : Efficiency of a reversible heat engine will be highest at $-273^{\circ} C$ temperature of cold reservoir.View Solution
Reason $R$ : The efficiency of Carnot's engine depends not only on temperature of cold reservoir but it depends on the temperature of hot reservoir too and is given as $\eta=\left(1-\frac{ T _2}{ T _1}\right)$.
In the light of the above statements, choose the correct answer from the options given below
- 9$Assertion :$ In isothermal process whole of the heat supplied to the body is converted into internal energy.View Solution
$Reason :$ According to the first law of thermodynamics : $\Delta Q = \Delta U + p\Delta V$ - 10A sample of an ideal gas undergoes an isothermal expansion. If $d Q, d U$ and $d W$ represent the amount of heat supplied, the change in internal energy and the work done respectively, thenView Solution
