An ideal gas occupies a volume of $2\, m^3$ at a pressure of $3\times10^6\, Pa$. The energy of the gas is
JEE MAIN 2019, Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1When $x$ amount of heat is given to a gas at constant pressure, it performs $x/3$ amount of work. The average number of degrees of freedom per molecule isView Solution
- 2The respective speeds of the molecules are $1, 2, 3, 4$ and $5\, km/sec.$ The ratio of their $r.m.s. $ velocity and the average velocity will beView Solution
- 3The temperature, at which the root mean square velocity of hydrogen molecules equals their escape velocity from the earth is closest to : [Boltzmans Constant $k_B = 1.38\times10^{-23}\, J / K$ Avogadro number $N_A = 6.02\times10^{26}\, / kg$ Radius of Earth $: 6.4\times10^6\, m$ Gravitation acceleration on Earth $= 10\, ms^{-2}$]View Solution
- 4Three particles have speeds of $2u$ , $10u$ and $11u$. Which of the following statements is correct?View Solution
- 5The speeds of $5$ molecules of a gas (in arbitrary units) are as follows : $2, 3, 4, 5, 6.$ The root mean square speed for these molecules isView Solution
- 6At a given temperature, the pressure of an ideal gas of density $\rho $ is proportional toView Solution
- 7A container with insulating walls is divided into two equal parts by a partition fitted with a valve. One part is filled with an ideal gas at a pressure $P$ and temperature $T$, whereas the other part is completely evacuated. If the valve is suddenly opened, the pressure and temperature of the gas will beView Solution
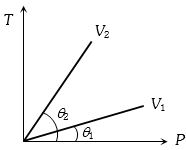
- 8From the following $P-T$ graph what interference can be drawnView Solution
- 9Two closed containers of equal volume filled with air at pressure $P_0$ and temperature $T_0$. Both are connected by a narrow tube. If one of the container is maintained at temperature $T_0$ and other at temperature $T$, then new pressure in the containers will beView Solution
- 10The r.m.s. speed of a group of $7$ gas molecules having speeds $(6, 4, 2, 0, -2, -4, -6)\, m/s$ is ...... $m/s$View Solution
