Three particles have speeds of $2u$ , $10u$ and $11u$. Which of the following statements is correct?
Diffcult
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1$N\,moles$ of a diatomic gas in a cylinder are at a temperature $T$. Heat is supplied to the cylinder such that the temperature remains constant but $n\,moles$ of the diatomic gas get converted into monoatomic gas. What is the change in the total kinetic energy of the gas ?View Solution
- 2An ideal gas has an initial pressure of $3$ pressure units and an initial volume of $4$ volume units. The table gives the final pressure and volume of the gas (in those same units) in four, processes. Which processes start and end on the same isothermView Solution
$\begin{array}{|c|c|c|c|c|} \hline & A & B & C & D \\ \hline P & 5 & 4 & 12 & 6 \\ \hline V & 7 & 6 & 1 & 3 \\ \hline \end{array}$
- 3View SolutionOne mole of a monoatomic ideal gas is mixed with one mole of a diatomic ideal gas. The molar specific heat of the mixture at constant volume is
- 4Same gas is filled in two vessels of the same volume at the same temperature. If the ratio of the number of molecules is $1: 4$, thenView Solution
$A.$ The $r.m.s$. velocity of gas molecules in two vessels will be the same.
$B.$ The ratio of pressure in these vessels will be $1: 4$
$C.$ The ratio of pressure will be $1: 1$
$D.$ The $r.m.s.$ velocity of gas molecules in two vessels will be in the ratio of $1: 4$
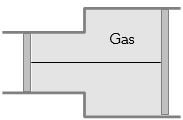
- 5View SolutionA gas is filled in the cylinder shown in the figure. The two pistons are joined by a string. If the gas is heated, the pistons will
- 6To what temperature should the hydrogen at room temperature $(27°C)$ be heated at constant pressure so that the $R.M.S.$ velocity of its molecules becomes double of its previous value ....... $^oC$View Solution
- 7A cylinder containing gas at $27\,^oC$ is divided into two parts of equal volume each $100\,^oc$ and at equal pressure by a piston of cross sectional area $10.85\,\, cm^2$. The gas in one part is raised in temperature to $100\,^oC$ while the other maintained at original temperature. The piston and wall are perfect insulators. How far will the piston move during the change in temperature.... $cm$ .View Solution
- 8For hydrogen gas ${C_p} - {C_v} = a$ and for oxygen gas ${C_p} - {C_v} = b$. So the relation between $a$ and $b$ is given byView Solution
- 9Five moles of helium are mixed with two moles of hydrogen to form a mixture. Take molar mass of helium $M_1=4\ g$ and that of hydrogen $M_2=2\ g$ If the internal energy of He sample of $100\,\,J$ and that of the hydrogen sample is $200\,\,J$, then the internal energy of the mixture is ..... $J$View Solution
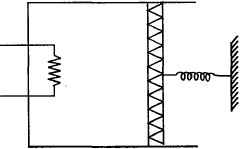
- 10An ideal monoatomic gas is confined in a cylinder by a spring loaded piston of cross section $8.0\times10^{-3}\, m^2$ . Initially the gas is at $300\, K$ and occupies a volume of $2.4\times10^{-3}\, m^3$ and the spring is in its relaxed state as shown in figure. The gas is heated by a small heater until the piston moves out slowly by $0.1\, m$. The force constant of the spring is $8000\, N/m$ and the atmospheric pressure is $1.0\times10^5\, N/m^2$ . The cylinder and the piston are thermally insulated. The piston and the spring are massless and there is no friction between the piston and the cylinder. The final temperature of the gas will be: (Neglect the heat loss through the lead wires of the heater . The heat capacity of the heater coil is also negligible)View Solution