The temperature, at which the root mean square velocity of hydrogen molecules equals their escape velocity from the earth is closest to : [Boltzmans Constant $k_B = 1.38\times10^{-23}\, J / K$ Avogadro number $N_A = 6.02\times10^{26}\, / kg$ Radius of Earth $: 6.4\times10^6\, m$ Gravitation acceleration on Earth $= 10\, ms^{-2}$]
JEE MAIN 2019, Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1Consider a $1\, c.c.$ sample of air at absolute temperature ${T_0}$ at sea level and another $1 cc$ sample of air at a height where the pressure is one-third atmosphere. The absolute temperature $T$ of the sample at that height isView Solution
- 2View SolutionIncrease in temperature of a gas filled in a container would lead to
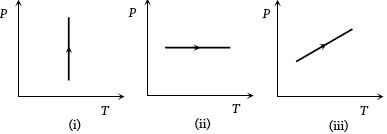
- 3View SolutionPressure versus temperature graphs of an ideal gas are as shown in figure. Choose the wrong statement
- 4The total internal energy of two mole monoatomic ideal gas at temperature $T=300\,K$ will be ...........$J$.View Solution
$\text { (Given } R =8.31\,J / mol.K )$
- 5The average translational energy and the $r.m.s.$ speed of molecules in a sample of oxygen gas at $300 K$ are $6.21 \times {10^{ - 21}}\,J$ and $484\, m/s$ respectively. The corresponding values at $600 K$ are nearly (assuming ideal gas behaviour)View Solution
- 6A mixture of ideal gases $7\,\, kg$ of nitrogen and $11 \,\,kg$ of $CO_2$. ThenView Solution
(Take $\gamma$ for nitrogen and $CO_2$ as $1.4$ and $1.3$ respectively)
- 7When one mole of monatomic gas is mixed with one mole of a diatomic gas, then the equivalent value of $\gamma$ for the mixture will be (vibration mode neglected)View Solution
- 8The Mean Free Path $\ell$ for a gas molecule depends upon diameter, $d$ of the molecule asView Solution
- 9Two containers $C_{1}$ and $C_{2}$ of volumes $V$ and $4 \,V$ respectively, hold the same ideal gas and are connected by a thin horizontal tube of negligible volume with a valve which is initially closed. The initial pressures of the gas in $C_{1}$ and $C_{2}$ are $p$ and $5 p$, respectively. Heat baths are employed to maintain the temperatures in the containers at $300 \,K$ and $400 \,K$, respectively. The valve is now opened. Select the correct statement.View Solution
- 10The air density at Mount Everest is less than that at the sea level. It is found by mountaineers that for one trip lasting a few hours, the extra oxygen needed by them corresponds to $30,000\, cc$ at sea level (pressure $1$ atmosphere, temperature $27°C$). Assuming that the temperature around Mount Everest is $-73°C$ and that the oxygen cylinder has capacity of $5.2 \,litre,$ the pressure at which ${O_2}$ be filled (at site) in cylinder is .... $atm$View Solution
