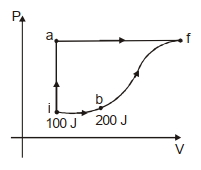
A thermodynamic system is taken form an initial state $i$ with internal energy $U_1=100 \ J$ to the final state along two different paths iaf and ibf, as schematically shown in the fire. The work done by the system along the paths $af$, ib and bf are $W _{ af }=200 \ J , W _{ ID }=50 \ J$ and $W _{ br }=100 \ J$ respectively. The heat supplied to the system along the path iaf, ib and bf are $Q_{\mid a t l} Q_{b r}$ and $Q_{10}$ respectively. If the internal energy of the sytem in the state $b$ is $U_b=$ $200 \ J$ and $Q_{l a t}=500 \ J$, the ratio $Q_{b J} / Q_{10}$ is:

IIT 2014, Advanced
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1Even Carnot engine cannot give $100\%$ efficiency because we cannotView Solution
- 2A carnot engine is made to work between $200\,^oC$ and $0\,^oC$ first and then between $0\,^oC$ and $-200\,^oC$. The ratio of efficiencies $\left( {\frac{{{\eta _2}}}{{{\eta _1}}}} \right)$ of the engine in two cases isView Solution
- 3In a thermodynamics process, pressure of a fixed mass of a gas is changed in such a manner that the gas releases $20 J$ of heat and $8J$ of work is done on the gas. If the initial internal energy of the gas was $30J.$ The final internal energy will be ...... $J$View Solution
- 4One mole of an ideal gas $(\gamma = 1.4)$ is adiabatically compressed so that its temperature rises from $27\,^oC$ to $35\,^oC$. The change in the internal energy of the gas is .... $J$ (given $R = 8.3 \,J/mole/K$)View Solution
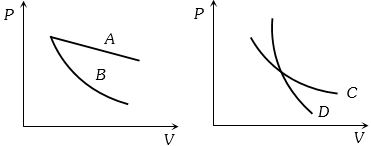
- 5In the following figure, four curves $A, B, C$ and $D$ are shown. The curves areView Solution
- 6In a certain thermodynamical process, the pressure of a gas depends on its volume as $kV ^{3}$. The work done when the temperature changes from $100^{\circ} C$ to $300^{\circ} C$ will be .......... $nR$, where $n$ denotes number of moles of a gas.View Solution
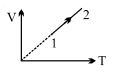
- 7An ideal gas undergoes the process $1 \rightarrow 2$ as shown in the figure, the heat supplied and work done in the process is $\Delta \,\,Q$ and $\Delta \,\,W$ respectively. The ratio $\Delta \,\,Q :$ $\Delta \,\,W$ isView Solution
- 8In a given process for an ideal gas, $dW = 0$ and $dQ < 0.$ Then for the gasView Solution
- 9One mole of an ideal monoatomic gas is compressed isothermally in a rigid vessel to double its pressure at room temperature, $27\,^oC$.The work done on the gas will beView Solution
- 10One mole of an ideal gas at $300 \mathrm{~K}$ in thermal contact with surroundings expands isothermally from $1.0 \mathrm{~L}$ to $2.0 \mathrm{~L}$ against a constant pressure of $3.0 \mathrm{~atm}$. In this process, the change in entropy of surroundings $\left(\Delta S_{\text {surr }}\right)$ in $\mathrm{J} \mathrm{K}^{-1}$ is $(1 \mathrm{~L} \mathrm{~atm}=101.3 \mathrm{~J})$View Solution