$\Delta Q=C_{P}^{n} \Delta T$
$\Delta U= C_{v}^{n} \Delta T$
$\Delta \omega=n\left(C_{P}-C_{N}\right) \Delta T$
$\triangle Q: \triangle \omega$
$\frac{C_{P}}{C_{V}}=\gamma,=\frac{C_{P}}{C_{P}-C_{V}}$
$=\frac{C_{P} / C_{V}}{C_{P} / C_{V}-1}=\frac{\gamma}{\gamma-1}$
Download our appand get started for free
Similar Questions
- 1$Assertion :$ When a glass of hot milk is placed in a room and allowed to cool, its entropy decreases.View Solution
$Reason :$ Allowing hot object to cool does not violate the second law of thermodynamics. - 2A diatomic gas $(\gamma=1.4)$ does $400 J$ of work when it is expanded isobarically. The heat given to the gas in the process is ............ $J$View Solution
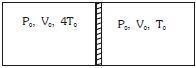
- 3Figure shows a cylindrical adiabatic container of total volume $2V_0$ divided into two equal parts by a conducting piston (which is free to move). Each part containing identical gas at pressure $P_0$ . Initially temperature of left and right part is $4T_0$ and $T_0$ respectively. An external force is applied on the piston to keep the piston at rest. Find the value of external force required when thermal equilibrium is reached. ( $A =$ Area of piston)View Solution
- 4The change in the entropy of a $1$ mole of an ideal gas which went through an isothermal process from an initial state $(P_1, V_1,T)$ to the final state $(P_2, V_2,T)$ is equal toView Solution
- 5During an adiabatic process, the pressure of a gas is found to be proportional to the cube of its temperature. The ratio of $\frac{{{C_P}}}{{{C_V}}}$ for the gas isView Solution
- 6$Assertion :$ The Carnot cycle is useful in understanding the performance of heat engines.View Solution
$Reason :$ The Carnot cycle provides a way of determining the maximum possible efficiency achievable with reservoirs of given temperatures. - 7A mixture of ideal gas containing $5$ moles of monatomic gas and $1$ mole of rigid diatomic gas is initially at pressure $P _0$, volume $V _0$ and temperature $T _0$. If the gas mixture is adiabatically compressed to a volume $V _0 / 4$, then the correct statement(s) is/are,View Solution
(Give $2^{1.2}=2.3 ; 2^{3.2}=9.2 ; R$ is gas constant)
$(1)$ The final pressure of the gas mixture after compression is in between $9 P _0$ and $10 P _0$
$(2)$ The average kinetic energy of the gas mixture after compression is in between $18 RT _0$ and $19 RT _0$
$(3)$ The work $| W |$ done during the process is $13 RT _0$
$(4)$ Adiabatic constant of the gas mixture is $1.6$
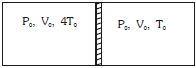
- 8Figure shows a cylindrical adiabatic container of total volume $2V_0$ divided into two equal parts by a conducting piston (which is free to move). Each part containing identical gas at pressure $P_0$ . Initially temperature of left and right part is $4T_0$ and $T_0$ respectively. An external force is applied on the piston to keep the piston at rest. Find the value of external force required when thermal equilibrium is reached. ( $A =$ Area of piston)View Solution
- 9This question has Statement $1$ and Statement $2.$ Of the four choices given after the Statements, choose the one that best describes the two Statements.View Solution
Statement $1 :$ An inventor claims to have constructed an engine that has an efficiency of $30\%$ when operated between the boiling and freezing points of water. This is not possible.
Statement $2:$ The efficiency of a real engine is always less than the efficiency of a Carnot engine operating between the same two temperatures.
- 10A gas is enclosed in a cylinder with a movable frictionless piston. Its initikl thermodynamic state at pressure $P_i=10^5 \mathrm{~Pa}$ and volume $V_i=10^{-3} \mathrm{~m}^3$ chanıes to i final state at $P_f=(1 / 32) \times 10^5 \mathrm{~Pa}$ and $V_f=8 \times 10^{-3} \mathrm{~m}^3$ in an adiabatic quasi-static process, such that $P^3 V^5=$ constant. Consider another thermodynamic process that brings the system from the same initial state to the same final state in two steps: an isobaric expansion at $P$, followed by an isochoric (isovolumetric) process at volume $V_f$. The amount of heat supplied to the system in the two-step process is approximatelyView Solution
