A gas is enclosed in a cylinder with a movable frictionless piston. Its initikl thermodynamic state at pressure $P_i=10^5 \mathrm{~Pa}$ and volume $V_i=10^{-3} \mathrm{~m}^3$ chanıes to i final state at $P_f=(1 / 32) \times 10^5 \mathrm{~Pa}$ and $V_f=8 \times 10^{-3} \mathrm{~m}^3$ in an adiabatic quasi-static process, such that $P^3 V^5=$ constant. Consider another thermodynamic process that brings the system from the same initial state to the same final state in two steps: an isobaric expansion at $P$, followed by an isochoric (isovolumetric) process at volume $V_f$. The amount of heat supplied to the system in the two-step process is approximately
IIT 2016, Advanced
In adiabatic process ideal gas equation: $PV ^{\vee}=$ Constant from given data, $P ^3 V^5=$ constant
So, $P V^{\frac{5}{3}}=k$
where, $Y=\frac{5}{3}$
Total work done during process $1$ and $2$,
$W_a=\frac{P_t V_t-P_i V_i}{1-y}$
$\rightarrow W_a=\frac{132 \times 10^{-5} \times 8 \times 10^{-3}-10^5 \times 10-3}{1-5 / 3}$
$W_a=112.5 J$
In adiabatic process the internal energy of system $\triangle Q=0$
$\text { so, } \Delta U=-W \Rightarrow-112.5 J$
Total heat supply between 1 and 2
$q_1+q_2=\Delta U+P_i\left(V_f-V_i\right)$
$=-112.5+10^5(8-1) \times 10^{-3}$
$=587.5 J$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A thermodynamic system is taken from state $A$ to $B$ along $ACB$ and is brought back to $A$ along $BDA$ as shown in the $PV$ diagram. The net work done during the complete cycle is given by the areaView Solution
- 2$5.6$ $liter$ of helium gas at $STP$ is adiabatically compressed to $0.7$ $liter$. Taking the initial temperature to be $T_1$, the work done in the process isView Solution
- 3Slope of isotherm for a gas (having $\gamma=\frac{5}{3}$ ) is $3 \times 10^5 \,N / m ^2$. If the same gas is undergoing adiabatic change then adiabatic elasticity at that instant is ........... $\times 10^5 N / m ^2$View Solution
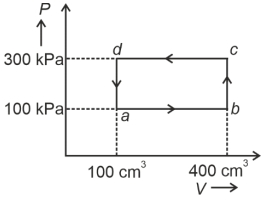
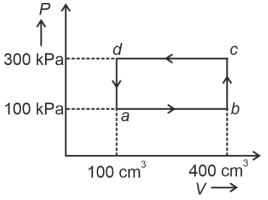
- 4A cyclic process of a thermodynamic system is taken through $a$ $b$ $c$ $d$ $a$. The work done by the gas along the path $b$ $c$ isView Solution
- 5$Assertion :$ The Carnot cycle is useful in understanding the performance of heat engines.View Solution
$Reason :$ The Carnot cycle provides a way of determining the maximum possible efficiency achievable with reservoirs of given temperatures. - 6A monoatomic gas of $n-$moles is heated from temperature $T_1$ to $T_2$ under two different conditions $(i)$ at constant volume and $(ii)$ at constant pressure. The change in internal energy of the gas isView Solution
- 7View SolutionAn ideal gas is compressed to half its initial volume by means of several processes. Which of the process results in the maximum work done on the gas?
- 8When an ideal gas in a cylinder was compressed isothermally by a piston, the work done on the gas was found to be $1.5 \times {10^4}\;joules$. During this process aboutView Solution
- 9A rigid diatomic ideal gas undergoes an adiabatic process at room temperature. The rational between temperature and volume for the process is $TV^x =$ constant, then $x$ isView Solution
- 10A cyclic process of a thermodynamic system is taken through $a$ $b$ $c$ $d$ $a$. The work done by the gas along the path $b$ $c$ isView Solution