An ideal gas, undergoing adiabatic change, has which of the following pressure temperature relationship?
AIPMT 1996, Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A frictionless heat engine can be $100 \%$ efficient only if its exhaust temperature is ............View Solution
- 2One mole of an ideal monoatomic gas undergoes the following four reversible processes:View Solution
Step $1$ It is first compressed adiabatically from volume $8.0 \,m ^{3}$ to $1.0 \,m ^{3}$.
Step $2$ Then expanded isothermally at temperature $T_{1}$ to volume $10.0 \,m ^{3}$.
Step $3$ Then expanded adiabatically to volume $80.0 \,m ^{3}$.
Step $4$ Then compressed isothermally at temperature $T_{2}$ to volume $8.0 \,m ^{3}$.
Then, $T_{1} / T_{2}$ is
- 3When a system is taken from state $i$ to state $f$ along the path $iaf$, it is found that $Q=50$ $cal$ and $W=20$ $cal$ Along the path $ibf\ Q = 36\ cal. \ W$ along the path $ibf$ is ....... $ cal$View Solution
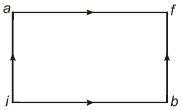
- 4A thermodynamic system is taken through the cycle $PQRSP$ process. The net work done by the system is ....... $J$View Solution
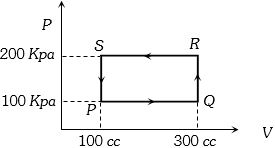
- 5On a $TP$ diagram, two moles of ideal gas perform process $AB$ and $CD$. If the work done by the gas in the process $AB$ is two times the work done in the process $CD$ then what is the value of $T_1/T_2$?View Solution
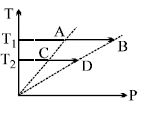
- 6View SolutionWhich of the following can be coefficient of performance of refrigerator?
- 7An engine is supposed to operate between two reservoirs at temperature $727°C$ and $227°C.$ The maximum possible efficiency of such an engine isView Solution
- 8A refrigerator is to maintain eatables kept inside at $9^{\circ} C .$ If room temperature is $36^{\circ} C$ calculate the coefficient of performance.View Solution
- 9The amount of work done in an adiabatic expansion from temperature $T$ to ${T_1}$ isView Solution
- 10Consider the efficiency of Carnot's engine is given by $\eta=\frac{\alpha \beta}{\sin \theta} \log _{e} \frac{\beta x}{k T}$, where $\alpha$ and $\beta$ are constants. If $T$ is temperature, $k$ is Boltzman constant, $\theta$ is angular displacement and $x$ has the dimensions of length. Then, choose the incorrect option.View Solution
