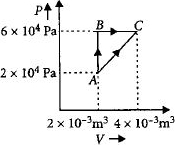
Figure below shows two paths that may be taken by a gas to go from a state $A$ to a state $C.$ In process $AB,$ $400 \,J$ of heat is added to the system and in process $BC,$ $100\, J$ of heat is added to the system. The heat absorbed by the system in the process $AC$ will be ...... $J$

AIPMT 2015, Diffcult
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1An ideal gas follows a process $PT =$ constant. The correct graph between pressure $\&$ volume isView Solution
- 2Two moles of helium gas are taken over the cycle $ABCDA$, as shown in the $P-T$ diagram. The net work done on the gas in the cycle $ABCDA$ is ...... $R$View Solution

- 3A system is provided with $200 \,cal$ of heat and the work done by the system on the surrounding is $40 \,J$. Then its internal energyView Solution
- 4$n\, moles$ of an ideal gas undergo a process $A \to B$ as shown in the figure. Maximum temperature of the gas during the process isView Solution
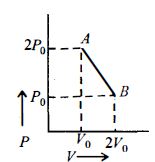
- 5View SolutionThe work done in which of the following processes is equal to the internal energy of the system?
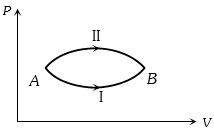
- 6A system goes from $A$ to $B$ via two processes $I$ and $II$ as shown in figure. If $\Delta {U_1}$ and $\Delta {U_2}$ are the changes in internal energies in the processes $I$ and $II$ respectively, thenView Solution
- 7The isothermal Bulk modulus of an ideal gas at pressure $P$ isView Solution
- 8View SolutionIn thermodynamics, heat and work are
- 9A gas is compressed at a constant pressure of $50N/{m^2}$ from a volume of $10{m^3}$ to a volume of $4{m^3}$. Energy of $100 J$ then added to the gas by heating. Its internal energy isView Solution
- 10View SolutionWhich of the following statements is correct for any thermodynamic system